|
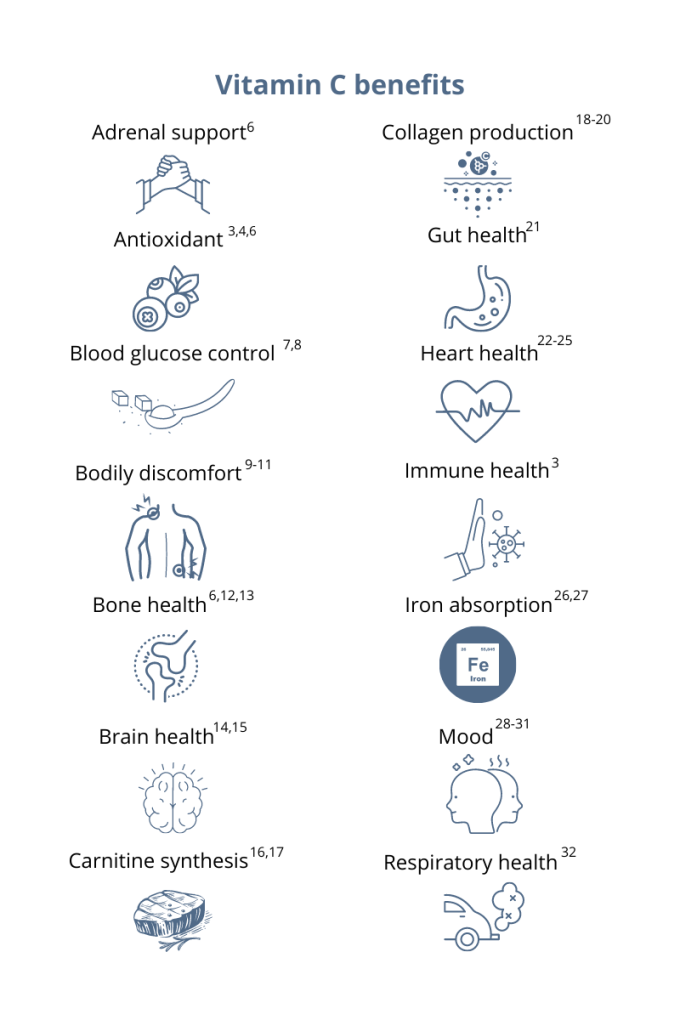
You’ve probably heard that vitamin C supports your immune system. This essential micronutrient seems to be everywhere! And it’s a good thing because, unlike most mammals, humans can’t synthesize vitamin C on their own.1 Also, vitamin C is water-soluble, which means the body quickly loses this essential vitamin through urine, so it’s important to make vitamin C a daily part of your diet.1 Having extremely low levels of vitamin C for prolonged periods can result in scurvy, a historical disease linked to pirates and sailors who faced long journeys at sea without fresh fruits and vegetables. While cases of scurvy in the United States are rare, a recent study reported that 31% of the US population are not meeting the daily recommended intake of vitamin C.1 Greater than 6% of the US population are severely vitamin C deficient, while low levels of vitamin C, associated with weakness and fatigue, were observed in 16% of Americans.2 As a whole, 20% of the US population showed marginally low levels of this essential micronutrient.2 How much vitamin C do I need?The US recommended daily dietary allowance of vitamin C is 75 mg for women and 90 mg for men.3 Experts recommend an estimated 200 mg of vitamin C daily for favorable health benefits.4 Adults can take up to 2,000 mg of vitamin C per day; however, high doses of vitamin C may cause diarrhea, nausea, and stomach cramps.5 Due to the varying health needs of individuals, it’s always a good idea to work with your healthcare practitioner to ensure that you are getting the right amounts of micronutrients in your daily diet. Where can you find this marvelous, multifaceted micronutrient? Ready to add vitamin C to your daily regimen? Talk to your healthcare practitioner about how much would be right for you.
References: 1. Granger M et al. Adv Food Nutr Res. 2018;83:281-310. 2. Schleicher RL et al. Am J Clin Nutr. 2009;90(5):1252-1263. 3. https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/. Accessed August 3, 2021. 4. Frei B et al. Crit Rev Food Sci Nutr. 2021;52(9):815-829. 5. Hathcock JN et al. AM J Clin Nutr. 2005;81(4)736-745. 6. Ashor AW et al. Eur J Clin Nutr. 2017;71(12):1371-1380. 7. Mason SA et al. Free Radic Biol Med. 2016;93:227-238. 8. Chen S et al. Clin J Pain. 2016;32(2):179-185. 9. Carr AC et al. J Transl Med. 2017;15(1):77. 10. Dionne CE et al. Pain. 2016; 157(11):2527-2535. 11. Chin KY et al. Curr Drug Targets. 2018;19(5):439-450. 12. Ratajczak AE et al. Nutrients. 2020;12(8):2263. 13. Dixit S et al. ACS Chem Neurosci. 2015;6(4):570-581. 14. Monacelli F et al. Nutrients. 2017;9(7):670. 15. Johnston CS et al. J of Nutr. 2007;137(7):1757–1762. 16. Johnston CS et al. Nutr Metab (Lond). 2006;3(35):1743-7075. 17. Moores J. Br J Community Nurs. 2013;Suppl:S6-S11. 18. Carr AC et al. Nutrients. 2017;9:1211. 19. Shaw G et al. Am J Clin Nutr. 2017;105(1):136-143. 20. Ratajczak AE et al. Nutrients. 2020;12(8):2263. 21. Ashor AW et al. Nutr Res. 2019;61:1-12. 22. Moser MA et al. Int J Mol Sci. 2016;17(8):1328. 23. Wu JR et al. J Cardiovasc Nurs. 2019;34(1):29-35. 24. Akolkar G et al. Am J Physiol Heart Circ Physiol. 2017;313(4):H795-H809. 25. Cook JD et al. Amer J Clin Nutr. 2001;73(1):93-98. 26. Saunders AV et al. Med J Aust. 2013;199(S4):S11-S16. 27. Amr M et al. Nutr J. 2013;12:31. 28. Consoli DC et al. J of neurochem. 2021;157(3):656-665. 29. Bajpai A et al. J Clin Diagn Res. 2014;8(12):CC04-CC7. 30. Koizumi M et al. Nutr Res. 2016;36(12):1379-1391. 31. Whyand T et al. Respir Res. 2018;19(1):79. 32. Azuma A et al. Tairyoku Kagaku Japanese J of Phys Fit and Sports Med. 2019;68(2):153-157.
0 Comments
Written by Julia Mellios
Your gut is one of the hardest working systems in your body. Meal after meal, it hustles to digest your food and extract the nutrients required to sustain you. Despite it’s solid work ethic, your gut can experience occasional performance hiccups, leading to digestive symptoms. While many of these are normal, others may indicate something more serious. Read on to discover what your symptoms say about your gut and ways to improve them. Normal, Everyday Symptoms Transient Belly Bloating What is it? Short-lived abdominal swelling due to trapped gas.
During and after meals, your gut produces enzymes and acids that break down your food, creating gas during this process. While bloating isn’t considered normal, it commonly occurs if you deviate from your standard diet, such as eating larger portions, rich and fatty meals or foods that are more laborious to breakdown, including excess carbohydrates and fibre. These place more burden on your digestive processes, subsequently increasing gas production and bloating. Flatulence
Borborymus
Symptoms That Require Attention Chronic Constipation
Diarrhoea
Malodorous Gas
Abdominal Pain
If you regularly experience one or more problematic symptoms, consult with a Natural Health Practitioner for further investigation, particularly if it is new or getting worse. Additionally, healthcare Practitioners have a suite of testing available to investigate the driver behind your symptoms. For instance, a stool test enables Practitioners to identify all of the bacteria and other microorganisms that inhabit your gut and how they contribute to your gut function and digestive symptoms. Ditch the Gut Glitches Don’t let your gut feelings bum you out! Instead, switch up your diet and lifestyle habits and rid yourself of symptoms such as bloating, gurgles and gas. If your gut symptoms are troubling you, speak with a natural healthcare Practitioner for a comprehensive plan to get you gut comfort, today. References1 Manichanh C, Eck A, Varela E, Roca J, Clemente JC, González A, et al. Anal gas evacuation and colonic microbiota in patients with flatulence: effect of diet. Gut. 2014 Mar;63(3):401-8. doi:10.1136/gutjnl-2012-303013. 2 Victoria State Government, Better Health Channel. Flatulence [Internet]. Melbourne VIC: Victoria State Government, Better Health Channel; 2014 [updated 2014 Aug; cited 2020 Jan 30]. Available from: https://www.betterhealth.vic.gov.au/health/ConditionsAndTreatments/flatulence. 3 Colledge NR, Walker BR, Ralston SH. Davidson’s principles and practice of medicine. 21st ed. Edinburgh (UK): Elsevier/Churchill Livingstone; 2010. p. 763-844. 4 Zhao Y, Yu YB. Intestinal microbiota and chronic constipation. Springerplus. 2016 Jul 19;5(1):1130. doi: 10.1186/s40064-016-2821-1. 5 Colledge NR, Walker BR, Ralston SH. Davidson’s principles and practice of medicine. 21st ed. Edinburgh (UK): Elsevier/Churchill Livingstone; 2010. p. 763-844. 6 NPS Medicinewise. What are the side effects of antibiotics? [Internet]. Sydney NSW: NPS Medicinewise; 2012 [updated 2017 Mar; cited 2020 Feb 18]. Available from: http://www.nps.org.au/medicines/infections-and-infestations/antibiotics/for-individuals/side-effects-of-antibiotics. Written by Rachel Baudistel Fermented foods and beverages, such as sauerkraut, kimchi and kombucha, were a regular part of our ancestors’ diets for thousands of years. Originally a method of preserving produce from harvest time through the cold days of winter;1 fermentation involves adding a bacterial or yeast starter to a food. These organisms convert starches and sugars to alcohol or acids, lengthening the food’s shelf life and producing the unique and tangy flavours we associate with fermented foods. Recently, these foods have seen a resurgence in popularity, not for their shelf lives but for their claimed digestive health benefits. As probiotics have also risen in popularity for similar reasons, let us explore how fermented foods stack up against a high quality probiotic. It All Begins In The Gut Our interest in strategies to improve digestive health has been fuelled by an expanding body of research indicating that poor gut health negatively affects many other body systems.2 Specifically, imbalances in the microbiome, the ecosystem of approximately 38 trillion bacteria and other organisms living in your digestive tract, have been connected not only to gut disorders but to mood, immune, inflammatory and autoimmune diseases, cancer and even cardiovascular disease.3,4 Research shows that taking live beneficial bacteria, such as probiotics, can improve microbiome health, reducing the symptoms of many health conditions,5,6 improving general health and lowering the risk of diseases such as those mentioned above.7 In light of this, probiotic supplements and fermented foods, both sources of potentially beneficial bacteria, have been put forward as effective options for improving microbiome health. Consequently, there is a misconception that they are interchangeable; however, there are fundamental differences between them. The Pros of Probiotics Probiotics are defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host”.8 Research has shown that specific types (strains) of probiotics, at defined doses, can help manage particular health conditions or symptoms, such as hayfever9 or bloating. To understand this further, let us use irritable bowel syndrome (IBS), a painful digestive disorder, as an example. (To read more about IBS, click here). Research indicates that the specific probiotic strain, Lactobacillus plantarum 299v, can relieve the bloating, intestinal pain and inflammation of medically diagnosed IBS.10,11 This benefit occurs when the probiotic is taken at a dose of 20 billion colony-forming units (CFU), which is a way of expressing the amount of live bacteria contained per probiotic capsule or dose of powder. Scientists give probiotic bacteria three names we can use to identify them and connect them to their health benefits: genus (e.g. Lactobacillus), species (e.g. plantarum), and strain (e.g. 299v). To get the health benefits associated with that strain, all three names must match the probiotic used in the scientific research, as well as the dose. Luckily, you can find information on strains and doses in a probiotic supplement just by reading the label. By choosing a probiotic from a reputable brand committed to quality, you can feel confident that you will receive:
So, if you are looking to improve a particular health condition, choose a strain-specific probiotic for best results. What’s the easiest way to find the right probiotic for your condition? See a Natural Healthcare Practitioner. Fermented Foods Vs Probiotics Although fermented foods contain live bacteria, the microbes responsible for fermentation do not confer the same health benefits as a probiotic supplement. In fact, an expert panel of scientists concluded that fermented foods have unidentified microbial content, meaning there is no guarantee of what bacterial strains or doses will be present in them. For this reason, they are fundamentally different from probiotics.12 That being said, many people experience non-specific digestive health benefits from including fermented foods in their diet, which could be related to the bacteria present, however, more research is needed to confirm this. Since fermentation breaks ingredients down into simpler parts, these foods are generally easier to digest, and fermentation also increases the nutritional value of the food.13 If you are healthy and do not require specific health benefits, fermented foods may be appropriate for you. Be aware that, while fermenting at home can be fun, it is not a risk-free process. Exposure to oxygen, for example, can allow mould, yeast and less beneficial bacteria to grow. High-quality probiotics, on the other hand, are produced under strict hygienic conditions that minimise the risk of introducing ‘bad’ microorganisms into your gut. If your digestive system is sensitive, you may be better off taking a probiotic. Choose What’s Right For You Fermented foods are a tasty addition to the diet and do appear to provide some non-specific digestive benefits to relatively healthy people, on the proviso that they are prepared correctly. However, for support with specific health conditions, it is important to choose a specific probiotic strain, at the right dose, for your condition. To access high-quality probiotics for your particular health needs, contact a Natural Healthcare Practitioner. References1 Chambers PJ, Pretorius IS. Fermenting knowledge: the history of winemaking, science and yeast research. EMBO Rep. 2010 Dec;11(12):914-20. doi: 10.1038/embor.2010.179. 2 Alam R, Abdolmaleky HM, Zhou JR. Microbiome, inflammation, epigenetic alterations, and mental diseases. Am J Med Genet B Neuropsychiatr Genet. 2017 Sep;174(6):651-660. doi: 10.1002/ajmg.b.32567. 3 Alam R, Abdolmaleky HM, Zhou JR. Microbiome, inflammation, epigenetic alterations, and mental diseases. Am J Med Genet B Neuropsychiatr Genet. 2017 Sep;174(6):651-660. doi: 10.1002/ajmg.b.32567. 4 Maranduba CM, De Castro SB, de Souza GT, Rossato C, da Guia FC, Valente MA et al. Intestinal microbiota as modulators of the immune system and neuroimmune system: impact on the host health and homeostasis. J Immunol Res. 2015;2015:931574. doi: 10.1155/2015/931574. 5 Niedzielin K, Kordecki H, Birkenfeld B. A controlled, double-blind, randomised study on the efficacy of Lactobacillus plantarum 299v in patients with irritable bowel syndrome. Eur J Gastro Hepat 2001;13:1-5. PMID: 11711768. 6 Costa DJ, Marteau P, Amouyal M, Poulsen LK, Hamelmann E, Czaaubiel M, et al. Efficacy and safety of the probiotic Lactobacillus paracasei LP-33 in allergic rhinitis: a double-blind, randomised, placebo-controlled trial (GA2LEN) study. Eur J Clin Nutr. 2014;1-6. doi: 10.1038/ejcn.2014.13. 7 Kalliomäki M, Salminen S, Arvilommi H, Kero P, Koskinen P, Isolauri E. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet. 2001 Apr 7;357(9262):1076-9. doi: 10.1016/S0140-6736(00)04259-8. 8 Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014 Aug;11(8):506-14. doi: 10.1038/nrgastro.2014.66. 9 Costa DJ, Marteau P, Amouyal M, Poulsen LK, Hamelmann E, Czaaubiel M, et al. Efficacy and safety of the probiotic Lactobacillus paracasei LP-33 in allergic rhinitis: a double-blind, randomised, placebo-controlled trial (GA2LEN) study. Eur J Clin Nutr. 2014;1-6. doi: 10.1038/ejcn.2014.13. 10 Niedzielin K, Kordecki H, Birkenfeld B. A controlled, double-blind, randomised study on the efficacy of Lactobacillus plantarum 299v in patients with irritable bowel syndrome. Eur J Gastro Hepat 2001;13:1-5. PMID: 11711768. 11 Ducrotté P, Sawant P, Jayanthi V. Clinical trial: Lactobacillus plantarum 299v (DSM 9843) improves symptoms of irritable bowel syndrome. World J Gastroenterol. 2012 Aug 14;18(30):4012-8. doi: 10.3748/wjg.v18.i30.4012. 12 Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat RevGastroenterol Hepatol. 2014 Aug;11(8):506-14.doi: 10.1038/nrgastro.2014.66. 13 Coyle D. What Is Fermentation? The Lowdown on Fermented Foods. https://www.healthline.com/nutrition/fermentation#what-it-is. Healthline. Accessed July 8, 2019. Written by Jo Herbert-Doyle
Did you know that your gut and your brain are in regular conversation? They have a pretty close relationship, with your mental state influencing gut function and vice versa. Perhaps you’ve experienced ‘butterflies in your stomach’, or felt nauseous before an event? Or felt your mouth water upon seeing or smelling a delicious meal? These are all very real phenomena and due to your mind-gut connection. Less ideal is how feeling under pressure or stressed decreases digestive function – this is because when your body-mind perceives ‘danger’ (which is what stress is from a physiological sense), then it’s not a priority to be eating, therefore digestion suffers and you may experience bloating or heartburn or some other uncomfortable symptom. Knowing this mind-gut link exists can help explain how feeling anxious may impact how your gut is functioning, but perhaps more significant (though not as well known) is that what’s going on in your gut could also be impacting your mental health. Lighting a Fire in the Brain Though the causes may be multifactorial, a key player in this situation is gut-derived inflammation. This can result from dysbiosis, which is an imbalance in your gut microbiota (the microbial life that resides within your intestines), and/or some degree of insult to the digestive tract lining – your gut ‘barrier’. Both your gut barrier and your microbiota work synergistically to support effective digestion, immune health, and overall wellbeing. However, if they become disrupted then biochemical signals or ‘cytokines’ are released that travel throughout your body causing what’s referred to as oxidative stress in any number of tissues in your body including your brain. Though many of these cytokines cannot cross what’s called the blood-brain-barrier, their presence triggers similar ‘alarm’ signals in the brain so it too is impacted by its own inflammatory processes. This sequence of events can be why symptoms such as poor mood, brain fog and anxiety occur; but for those who want a more specific example – inflammation can promote the neurotransmitter precursor tryptophan to produce quinolinic acid1 instead of serotonin and melatonin – the former situation leading to what can most simply be described as nerve cell ‘agitation’ that can present as anxiety.2 Soothe Your Gut to Settle Your MindAs gut health is fundamental to all aspects of wellbeing then any symptoms really need to be attended to – and to do that with the least guesswork then my recommendation is to see a natural healthcare Practitioner who can help you navigate what may be the contributing causes for you, e.g. do you need to tweak your diet? Get some help with digesting certain foods? Heal your gut barrier? Equally, if you are struggling with stress, sleep, or experiencing regular mood problems then speak to your Practitioner about this too – there are many natural medicine solutions available, but to find the best ones for you will require assessing your personal situation first. Is it too Early to Rely on Psychobiotics? If you are already a fan of natural medicine you may be wondering what the role of probiotics are in this, particularly as there is emerging talk of ‘psychobiotics’ or probiotics that can impact mood. This is clearly of interest to those who may be experiencing stress or anxiety, so I’d say to you that the human evidence for what may be the ‘best’ probiotic strain and at what dose to help is still in its infancy; but watch this space for future research developments. What we do know currently is that supporting whole gut health by decreasing gut-derived inflammation and increasing the diversity of the gut microbiota is a proven clinical approach to whole-body wellbeing – and this includes improving mental health. So if you experience symptoms impacting either your mind or your gut, please don’t put up with them any longer, or try to figure it all out on your own. Contact your Clinical Naturopath or Integrative Doctor and let them know what’s going on, so they can personalise a solution for your situation. References1 Lugo-Huitrón R, Muñiz PU, Pineda B, Pedreaza-Chaverri J, Rios C, Pérez-de la Cruz V. Quinolinic acid: an edogenous neurotoxin with multiple targets. Oxidative Medicine and Cellular Longevity. 2013;Article ID 104024, 14 pages. Available from: https://www.hindawi.com/journals/omcl/2013/104024/ 2 Bryleva EY, Brundin L. Kynurenine pathway metabolites and suicidality. Neuropharmacology. 2017 Jan;112(Pt B):324-330. doi: 10.1016/j.neuropharm.2016.01.034. Epub 2016 Jan 26. Available from: https://www.sciencedirect.com/science/article/pii/S0028390816300338 Written by Gauri Yardi
What do a cold, depression, and hayfever have in common? If you said “they’re all health conditions”, or even “they’re all inflammatory health conditions”, you would be right. However, there is something more unusual that connects the three. Give up? All three are influenced by your gut microbiome, the microorganisms that call your digestive tract ‘home’. You may be wondering how these tiny gut inhabitants could have any bearing on your throat, joints, and/or brain. In this article, we will find out how your gut influences these seemingly unrelated areas, as well as how to prevent your gut from making you sick, sad or inflamed. Cold-Busting Colleagues: Your Gut and Immune System Work Hand-in-Hand Your immune system’s main job is to defend you from pathogens (disease-causing microorganisms). Since pathogens are typically inhaled or swallowed, it makes sense for the immune system to concentrate on your respiratory and digestive tracts. In fact, 70% of the immune system is housed in your gut.1 It lies beneath the lining of your intestines, ready to spring into action if a pathogen enters your gut, to try to prevent you getting sick. By contrast, some bacteria have a positive influence on your immune system. A healthy gut microbiome interacts with the intestinal immune system in ways that increase your body’s immune defences. However, a microbiome out of balance, which does not contain high levels of beneficial bacteria, is less likely to help you resist infection, including colds and flu (click here to read more about what might upset your gut microbiome). Fortunately, certain strains (types) of probiotic bacteria improve the bacterial balance in your gut, with beneficial flow-on effects for your immune system. Lactobacillus rhamnosus (LGG®),2 Lactobacillus paracasei (8700:2) and Lactobacillus plantarum (HEAL 9)3 all stimulate the immune system and improve resistance to infection. In fact, the combination of 8700:2 and HEAL 9 has been shown to reduce the severity and duration of common cold symptoms.4 If you struggle with frequent colds and flu, working with a natural healthcare Practitioner to strengthen your gut microbiome may help. A healthy gut microbiome interacts with the intestinal immune system in ways that increase your body’s immune defences. However, a microbiome out of balance, which does not contain high levels of beneficial bacteria, is less likely to help you resist infection, including colds and flu (click here to read more about what might upset your gut microbiome). Jumping at Shadows: The Overactive Immune System Another possible consequence of poor gut bacterial balance is inflammation, a key feature of autoimmune (e.g. rheumatoid arthritis) and allergic disease (e.g. hayfever). In these conditions, the immune system misidentifies harmless substances as threats, and launches an immune response against them. The resulting inflammation creates the symptoms you associate with allergy and autoimmunity, e.g. a blocked nose and watering eyes in hayfever, or joint pain and swelling in rheumatoid arthritis. Fortunately, certain probiotic strains, namely LGG® and Lactobacillus paracasei (LP-33®), can stimulate your immune system to produce anti-inflammatory compounds, reducing inflammation and symptoms. For example, research in hundreds of people has shown that LP-33® significantly improves hayfever symptoms.5,6 Interestingly, LGG®, when taken during pregnancy and breastfeeding, can reduce the incidence of eczema (an inflammatory skin disease) in children, by supporting the healthy development of the gut microbiome and the immune system.7 If your immune system is in overdrive, make an appointment with a natural healthcare Practitioner to help bring it back into line. Gut Feelings: How Bacteria Make or Break Your MoodMore and more research is supporting an unexpected cause of depression: inflammation. Specifically, inflammation throughout the body (known as systemic inflammation), and even inflammation of the brain, may contribute to depression. As you have already learned, the interaction between bad gut bacteria and the immune system can cause inflammation. However, did you know that the inflammatory chemicals released within your gut can also cause an inflammatory response in your brain? If gut inflammation can influence mood, you may be wondering if specific probiotics can improve mood or reduce the symptoms of depression. While this is a hot topic in scientific research, we do not currently know which specific probiotic strains can influence mood. However, a good start in supporting healthy mood is taking steps to reduce inflammation in the body. What we do know is maximising your gut health, e.g. by eating plenty of fibre-rich wholefoods (to provide your gut bacteria with their preferred food), can also increase the numbers of good bacteria, which is the best way to influence your mood via your gut. If your bacterial balance has become disrupted due to a stomach bug, antibiotics, or other causes, strains which support beneficial bacteria, such as LGG®, Saccharomyces cerevisiae var. boulardii, and Bifidobacterium animalis ssp lactis (BB-12®) may help improve the composition of your gut microbiome. Great Health is All in the Gut By interacting with your immune system, your gut bacteria influences your ability to resist infection, reduce inflammation, and maintain a healthy mood. If you are wondering whether your gut may be making you sick, sad or inflamed, make an appointment with a natural healthcare Practitioner today. Together, you can assess your bacterial balance, and make a plan to improve your specific symptoms. References1 Gill HS. Probiotics to enhance anti-infective defences in the gastrointestinal tract. Best Pract Res Clin Gastroenterol. 2003;17(5):755-773. doi: 10.1016/s1521-6918(03)00074-x. 2 Feleszko W, Jaworska J, Rha RD, Steinhausen S, Avagyan A, Jaudszus A, et al. Probiotic-induced suppression of allergic sensitization and airway inflammation is associated with an increase of Tregulatory-dependent mechanisms in a murine model of asthma. Clin Exp Allergy. 2007;37(4):498-505. doi: 10.1111/j.1365-2222.2006.02629.x. 3 Lavasani S, Dzhambazov B, Nouri M, Fåk F, Buske S, Molin G, et al. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLoS One. 2010; 5(2): e9009. doi: 10.1371/journal.pone.0009009. 4 Berggren A, Lazou Ahrén I, Larsson N, Önning G.. Randomised, double-blind and placebo-controlled study using new probiotic lactobacilli for strengthening the body immune defence against viral infections. Eur J Nutr 2011; 50(3):203-210. doi: 10.1007/s00394-010-0127-6. 5 Costa DJ, Marteau P, Amouyal M, Poulsen LK, Hamelmann E, Czaaubiel M, et al. Efficacy and safety of the probiotic Lactobacillus paracasei LP-33 in allergic rhinitis: a double-blind, randomised, placebo-controlled trial (GA2LEN) study. Eur J Clin Nutr. 2014 May;68(5):602-7. doi: 10.1038/ejcn.2014.13. 6 Peng G-C, Hsu C-H. The efficacy and safety of heat-killed Lactobacillus paracasei for treatment of perennial allergic rhinitis induced by house-dust mite. Pediatr Allergy Immunol. 2005 Aug;16(5):433-438. doi: 10.1111/j.1399-3038.2005.00284.x. 7 Kalliomäki M, Salminen S, Arvilommi H, Kero P, Koskinen P, Isolauri E. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet. 2001;357(9262):1076-9. By Melissa Blake, ND
Our bodies have an amazing natural ability of keeping to a daily schedule via an internal 24-hour master clock.1 This clock contributes to the patterns, also known as circadian rhythms, of many biological activities including sleep-wake cycles, eating patterns, and hormone function.1 Finding ways to support and balance this clock, along with the many systems it regulates, may offer a novel way to optimize health. One way to optimize is through diet timing. The circadian diet as a way of eating takes into account not only what we eat, but when.2 It is an approach to eating that synchronizes food intake around our biological clocks, emphasizing eating in sync with the body’s natural tendencies and instincts. This means eating during daylight hours or hours when we are naturally more active. Eating in this way can help support circadian rhythms and contribute to overall health and wellness.2 As diets and terms including intermittent fasting, circadian diet, and time-restricted eating gain popularity, the question may arise as to whether the principle on which this “circadian approach” applies to other aspects of nutrition, including supplementation. 5 common supplements Although there’s still much to learn about optimal timing for both food and supplements, current evidence suggests it may play a role.3 Here are a few general guidelines for five common supplements to help you add the extra layer of timing and optimize your plan: B complex B vitamins are often recommended to support healthy energy and mood.4 There is some evidence that taking B vitamins before bed can have a negative effect on sleep quality.5 Consider taking any B vitamins, including a B complex, earlier in the day with food. Fish oil The most common complaints I hear about fish oil are burping or nausea. Taking fish oil supplements with food, divided into two doses, may help reduce these harmless yet annoying side effects. Magnesium Magnesium is an essential micronutrient that plays a role in hundreds of reactions in the body.6 Due to the overall benefits of magnesium supplementation, consistency is more important than any timing in this case. Known for muscle relaxation and improved sleep, taking magnesium before bed may enhance those benefits in some people.7 Others may notice digestive issues and may choose to take with food. Probiotics Any recommendation related to probiotic supplementation should be based on the specific strain; however, much of this detailed evidence does not yet exist. Meal timing has more or less of an impact on probiotics depending on the strain, the dose, delivery method, etc.8 The consensus, however, is to take probiotics 30 minutes before or during a meal versus after eating.9 Another guideline is to space probiotics away from antibiotic medications by two hours to reduce interaction. Vitamin D Along with vitamins A, E, and K, vitamin D is a fat-soluble vitamin that is better absorbed when taken with a fat-containing meal (ex. fatty fish, avocado, olive oil, cheese, eggs).10 Although we do not have substantial evidence to support specific timing, it may make sense to take vitamin D supplements in the morning with breakfast to mimic the timing of exposure to natural sunlight. Summary As we continue to learn about specific supplements and optimal timing, consider that the best timing is the one you can stick with. You cannot benefit from a supplement you do not take. The most important thing is to take your supplements at a time that is convenient for you so you can be consistent. Work with a knowledgeable healthcare provider to determine the optimal plan for you that includes quality, quantity, and timing. References:
Feeling off? An unbalanced gut microbial community, also known as dysbiosis, can wreak havoc on your health. Your gut is home to around 100 trillion microbes, and most of these little critters are good for you.1 However, not all microbes provide the same health benefits. When potentially harmful bacteria have stronger effects than your good bacteria, it can negatively affect your health.
Here are 7 signs that your gut could use some love: Acne Has your skin been suffering from breakouts? An unbalanced gut might be the culprit. Recent studies have revealed a link between reduced bacterial diversity in the human gut and acne.2 Acne affects up to 50 million people in the United States and is the nation’s most common skin condition.3 What can you do to fight back against stubborn blemishes? Take a close look at your diet. Research has identified a link between the Standard American Diet (SAD) and acne.2 An overabundance of saturated fats, dairy products, and refined carbohydrates leaves Americans undernourished and can contribute to the development of blemishes.2 Omega-3 fatty acids are also in short supply in the SAD; supplementation with omega-3s has been shown to help reduce acne.4 Mood Feeling down? The bacteria living in your gut could be partially to blame. You’ve probably heard of the gut-brain connection; many studies have shown a relationship between mood and the gut.5 Via the vagus nerves, your thoughts and moods send signals to your digestive tract (and immune system) and vice versa.6 Recent studies suggest that a high-quality probiotic may help to improve mood and cognition.7-10 GI upset Too many trips to the porcelain throne? Losing friends to flatulence? Confounded by occasional constipation? These annoying symptoms could be signs of dysbiosis.11 To address these issues, a good place to start is to look at your medications and supplements to see if any may cause occasional diarrhea or constipation. Additionally, an elimination diet can be a good way to help determine if certain foods might be contributing to your symptoms. Your poop could be telling you a lot about your health, so keeping tabs on your bathroom visits is important. Immune system 70% of your immune system resides in your gut.12 A healthy balance of gut bacteria helps support a sufficient immune response. Sleep Are you counting sheep by the thousands, yet sleep is still elusive? The critters living in your gut could be partially to blame. A recent study showed that better and longer sleep is associated with microbiome diversity.13 Sugar cravings If you often find yourself craving sugary treats, this could be partially due to the microbes in your gut.14 For example, the bacteria in your gut may signal that they would like a bacon maple doughnut, even though you planned to have a salad instead. The most effective way to turn down the volume on the signals for sugary snacks? Starve those nasty bugs by changing up your daily diet. You can improve your guts microbiome by favoring vegetables, fruits, and fermented foods. Over time the cravings can diminish. Weight fluctuations Are you struggling to maintain a healthy weight? The bacteria in your gut could be a major player. Everyone’s microbiome is unique—made up of various strains of bacteria and protozoa (a group of single-celled microorganisms)—some of these gut dwellers have been shown to influence weight and even overall health. If you are experiencing sudden weight loss or unusual changes in your bowel habits, you should contact your healthcare practitioner. 7 ways to improve gut health Diet The more diverse your diet, the more diverse your microbiome! Eat the rainbow by loading up on a variety of different-colored fruits and vegetables, whole grains, and lean protein sources like fish and legumes. Another great way to support your gut is by eating fermented foods. Building a rich microbiome is a fantastic way to care for your health and immune system. Additionally, here are 5 foods to reduce to support your gut health. Probiotics High-quality probiotics can be a fantastic way to support your gut health. When you have a rich and diverse “forest” of good bacteria, the harmful bacteria are crowded out. This increase of good bacteria and the resulting reduction in harmful bacteria can improve your digestion and ability to better absorb macronutrients and micronutrients. Two important things to remember when selecting a probiotic: 1. Different strains for different gains, meaning different probiotic strains support specific health goals. 2. Not all probiotics are created equal. Here are six things to look for in a probiotic. Prebiotics Remember to feed your good bacteria! The good bacteria in your gut live on prebiotics. Prebiotics resist digestion so that they can reach the good bacteria living in your colon. Your good bacteria then ferment (eat) the prebiotics. So, probiotics and prebiotics go together like…well, probiotics and prebiotics! Supplements Many supplements are formulated to support a healthy gut and a strong digestive tract. Some great options are glutamine, inner leaf aloe, and zinc-carnosine. Additionally, recent studies have shown the benefits of vitamins C, B2 (or riboflavin), and D on the digestive tract and microbiota of the human gut.15-17 Exercise Need another reason to exercise? Studies have shown that exercise can increase microbiota diversity within the gut.18 So find your favorite way to break a sweat and get moving. Your gut will thank you! Developing a yoga practice may be a great place to start, as studies have shown that yoga has proven benefits for relieving intestinal distress.19-20 Hydrate Drink up; the gut’s mucosal lining is 98% water!21 Water not only helps to expel toxins through urine and stool, but it also helps to prevent occasional constipation. It’s important to support the mucosal lining of your gut; this lining helps shield your gut’s inner epithelial walls from undesirable microbes.21 Relax The gut and brain are in constant contact via the vagus nerves. Relaxation has been found to positively impact gut health.22 If you think your gut may be the source of your symptoms or just needs some love, reach out to your healthcare practitioner to create a plan of action to reach your best gut health. Unusual GI symptoms can be a sign of a more serious health concern. References
Many of us have learned to “trust our gut.” But if we ask most people what our digestive tract is for, they’ll probably explain it as a way of absorbing nutrients and disposing of waste. So where do phrases such as “trusting our gut” or “unable to stomach the results” come from? It turns out there is a two-way street between our digestive organs and our brains. What we eat and the state of our digestive tract can have a profound impact on our moods and behaviors. And probably unsurprisingly at this point, our gut microbiome is also involved.
Our vagus nerves, which control much of our “rest and digest” response, make a direct connection from our brains to our digestive tracts. Incidentally, these nerves also innervate our heart and communicate with our immune systems.1 These nerves help our brains communicate to our stomachs to start releasing stomach acid and digestive enzymes, and they carry signals from our digestive organs that tell our brains whether we’re hungry or full.1 But there’s a whole lot more to the story than these basic logistical messages. Our thoughts and moods are sending signals to our digestive tract (and immune system) and vice versa.1 Have you ever felt nausea from a stressful situation or lost your appetite from bad news? That message was carried from your brain to digestive tract via your vagus nerve.2 What might be news is that the situation in your digestion can also send signals to your brain.2 Is your digestive system reacting with something you ate? That signal is conveyed to your brain and can color the rest of your day or night.2 And to add another layer of complexity, the bacteria and other organisms in our gut can make or simulate neuroactive molecules like gamma-aminobutyric acid (GABA), melatonin, and serotonin.2 As you may know, these substances can make a big impact on our mood and how alert we feel. And stress can change the number of healthy bacteria (such as lactobacilli) in our guts and weaken the digestive tract and blood-brain barriers—letting molecules and even bacteria in that would be excluded under normal conditions.2,3 Conversely, consuming some probiotic species can improve our mood.2 This is all to say, everything is connected. I’ve seen many patients with digestive troubles (myself included) who’ve been told by someone, “It’s all in your head.” This is an oversimplified message, because stress and emotions absolutely play a role in digestive health, and the opposite is also true. Likewise, many patients who come in for issues with their mood have related concerns in their digestion that must be addressed before they see significant progress. Often when it comes to health, the symptoms can lead to the root of the problem, but only when you understand the connections. References:
Did you know that the majority of your immune system is in your gut?1 Picture a lush, harmonious rainforest teeming with diverse forms of life; this is what a healthy gut should look like at a microbial level. With more than 1,000 microbial species, most of which are bacteria, your gut’s microbiome makes up an essential part of your immune system.2 The good bacteria in your gut protect you from pathogenic invaders and keep harmful bacteria from growing out of control. The good bacteria that live in your gut are an essential part of your body’s microbiome. Here are 10 of the best ways to increase your good gut bacteria.
1. Eat whole foods The human body is not meant to run on food-like substances; diet matters! Eating processed foods and refined sugars starves the good bacteria in your gut, allowing harmful bacteria to grow out of control, also known as dysbiosis.3,4 Instead of filling up on processed foods, eat the rainbow. Choosing to eat a wide variety of whole high-fiber foods feeds your good bacteria and, in turn, nourishes your microbiome. Choosing organic produce is also a great way to add good bacteria to your gut’s ecosystem.5 By keeping your good bacteria strong and healthy, they can keep you healthy too. 2. Eat fermented foods The creation of fermented foods dates back thousands of years, as far as 10,000 BCE, with the advent of yogurt; the health benefits of yogurt are described in Ayurvedic scripts from 6,000 BCE.6 Fermented foods are a great way to support your health by adding good bacteria to your gut’s population of roughly 100 trillion bacteria and microbes. Here are some tasty options for adding fermented foods to your daily diet:
Probiotics are “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.”7 Choosing a high-quality clinically supported probiotic is a great way to introduce some new friends to your gut. Different strains of probiotics offer different health benefits; here are six things to look for in a probiotic. 4. Feed your good bacteria Now that you’ve loaded up on good bacteria from probiotic foods, it’s time to feed your new friends! Probiotics and prebiotics go together like, well, probiotics and prebiotics! Prebiotics are fibers that can resist the digestive process; once prebiotics reach the colon, they are “eaten” (selectively fermented) by specific strains of friendly gut bacteria.8,9 Here are some whole foods filled with prebiotics to feed your microscopic friends:10
Research shows a relationship between sleep and diversity within the gut microbiome.11 Consistent, uninterrupted sleep of between seven to nine hours per night has been shown to have a positive correlation to bacterial strains related to good health.12,13 6. Get moving Consistent exercise results in increased good bacteria. In a six-week study, exercise was shown to increase the numbers of certain strains of good bacteria.14 However, when followed by a six-week sedentary period, the increase of microbiota returned to baseline, so it’s important to get moving and stay moving.14 7. Destress Stress has been shown to have a negative impact on gut health. Heightened stress levels can trigger an increase in bad/pathogenic bacteria, crowding out good bacteria.15,16 Try these six simple ways to reduce stress. 8. Get some vitamin D A 2019 study found that skin exposure to UVB light positively impacts the diversity and composition of the human microbiome.17 Vitamin D supplementation also has positive impact on the gut’s microbiome by increasing bacterial diversity and richness.”18,19 9. Take it easy on the cocktails It’s well known that red wine can support good health, but too much alcohol has been shown to have a negative impact on good gut bacteria.20,21 The CDC 2020-2025 guidelines recommend one drink or less in a day for women and two drinks or less in a day for men.22 The CDC guidelines also caution against beginning drinking for possible health benefits and advise that “drinking less is better for health than drinking more.”22,23 The health benefits of red wine likely stem from polyphenols from the grape skins present in the wine-making process.24-27 Polyphenols are complex compounds found in fruits and vegetables that protect plants from pathogens and UV radiation; these compounds act as an antioxidant and as a prebiotic in the human body.28,29 Plenty of foods are packed with polyphenols, such as:30
Smoking causes a decrease in microbiome diversity; smoking withdrawal has been found to increase gut microbial diversity.31 Quitting smoking is a great way to show your little critters some love.31 All of these are great ways to help increase the friendly bugs working hard in your gut. Which will you try first? References: 1. Vighi G et al. Allergy and the gastrointestinal system. Clin Exp Immunol. 2008;153 Suppl 1(Suppl 1):3-6. 2. Wang W et al. Gut microbiota and allogeneic transplantation. J Transl Med. 2015;13:275. 3. Gagliardi A et al. Rebuilding the gut microbiota ecosystem. Int J Environ Res Public Health. 2018;15(8):1679. 4. Di Rienzi SC et al. Adaptation of the gut microbiota to modern dietary sugars and sweeteners. Adv Nutr. 2020;11(3):616-629. 5. Blum WEH et al. Does soil contribute to the human gut microbiome? Microorganisms. 2019;7(9):287. 6. Fisberg M et al. History of yogurt and current patterns of consumption. Nutr Rev. 2015;73 Suppl 1:4-7. 7. Hill C et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506-514. 8. Charalampopoulos D et al. Prebiotics in foods. Curr Opin Biotechnol. 2012;23(2):187-191. 9. Gibson GR et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14(8):491-502. 10. Jovanovic-Malinovska R et al. Oligosaccharide profile in fruits and vegetables as sources of prebiotics and functional foods. Int J Food Prop. 2014;17(5):949-965. 11. Smith RP et al. Gut microbiome diversity is associated with sleep physiology in humans. PLoS One. 2019;14(10):e0222394. 12. Fei N et al. Gut microbiota alterations in response to sleep length among African-origin adults. PLoS One. 2021;16(9):e0255323. 13. Bowers SJ et al. Repeated sleep disruption in mice leads to persistent shifts in the fecal microbiome and metabolome. PLoS One. 2020;15(2):e0229001. 14. Mailing LJ et al. Exercise and the gut microbiome: a review of the evidence, potential mechanisms, and implications for human health. Exerc Sport Sci Rev. 2019;47(2):75-85. 15. Zeng MY et al. Mechanisms of inflammation-driven bacterial dysbiosis in the gut. Mucosal Immunol. 2017;10(1):18-26. 16. Kiecolt-Glaser JK et al. Modulation of cellular immunity in medical students. J Behav Med. 1986;9:5–21. 17. Bosman ES et al. Skin exposure to narrow band ultraviolet (UVB) light modulates the human intestinal microbiome. Front Microbiol. 2019;10:2410. 18. Bashir M et al. Effects of high doses of vitamin D3 on mucosa-associated gut microbiome vary between regions of the human gastrointestinal tract. Eur J Nutr. 2016;55(4):1479-1489. 19. Waterhouse M et al. Vitamin D and the gut microbiome: a systematic review of in vivo studies. Eur J Nutr. 2019;58(7):2895-2910. 20. Zhang X et al. Alcohol-induced changes in the gut microbiome and metabolome of rhesus macaques. Psychopharmacology. 2019;236(5):1531-1544. 21. Dubinkina VB et al. Links of gut microbiota composition with alcohol dependence syndrome and alcoholic liver disease. Microbiome. 2017;5(1):141. 22. https://www.cdc.gov/alcohol/fact-sheets/moderate-drinking.htm. Accessed September 21, 2021. 23. https://www.dietaryguidelines.gov/sites/default/files/202012/Dietary_Guidelines_for_Americans_2020-2025.pdf. Accessed September 21, 2021. 24. https://journalsblog.gastro.org/is-red-wine-consumption-good-for-your-intestinal-microbiome/. Accessed September 21, 2021. 25. https://www.winespectator.com/articles/understanding-wine-polyphenols-health-benefits. Accessed September 21, 2021. 26. Wiciński M et al. The influence of polyphenol compounds on human gastrointestinal tract microbiota. Nutrients. 2020;12(2):350. 27. Cavallini G et al. Resveratrol requires red wine polyphenols for optimum antioxidant activity. J Nutr Health Aging. 2016;20(5):540-545. 28. Pandey KB et al. Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev. 2009;2(5):270-278. 29. Nazzaro F et al. Polyphenols, the new frontiers of prebiotics. Adv Food Nutr Res. 2020;94:35-89. 30. Yang J et al. Polyphenols in foods. Nutri Today. 2016;51(6):290-300. 31. Capurso G et al. The interaction between smoking, alcohol and the gut microbiome. Best Pract Res Clin Gastroenterol. 2017;31(5):579-588. As women, we’re constantly seeking balance. Balancing our families and careers, time and energy, work and fun can be a struggle. But balancing that delicate microbiome in our guts doesn’t have to be a challenge, thanks to probiotics. Probiotics are “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.”1 There are many myths about probiotics, but the health benefits aren’t one of them. If you’re curious why you should consider taking a daily probiotic and how it can benefit you, consider the following six reasons. 1. Probiotics support immune health. It’s a well-known stereotype that women tend to put others first. The downside of this is that we don’t always take the time to take care of ourselves. The gut plays an active role in immune health, with intestinal bacteria helping to regulate immune cell activity.2 Taking a daily probiotic may be helpful for supporting immune function.3 2. Probiotics can provide self-care for “down there.” Yes, that “down there.” The vagina is an ecosystem that requires a delicate balance, just like the gut. Luckily, there are clinically researched probiotic strains that have been shown specifically to impact feminine health.4 Two such probiotic strains are Lactobacillus rhamnosus GR-1® and Lactobacillus reuteri RC-14®, which work by traveling through the digestive tract to the vagina.5 Research shows that once there, the two probiotic strains work to help maintain a healthy vaginal environment by increasing the number of good bacteria.4,6,7 3. Certain probiotic strains support weight maintenance. Most people know that a healthy weight correlates to a healthy life.8 But did you know that probiotics can help you maintain that healthy weight? One probiotic strain in particular, Bifidobacterium lactis B-420™, has shown in clinical research that it can help weight maintenance by controlling body fat.9 4. Probiotics support gut health. Women aren’t the only ones whose guts benefit from daily probiotics; this one’s for everyone, even kids! Much research has been done on the benefits of probiotics on gut and digestive health, so it’s no wonder it’s a common reason practitioners recommend this first line therapy for patients seeking digestive support. Look for strains like Bifidobacterium lactis Bi-07® and Lactobacillus acidophilus NCFM®, both of which have been researched for their relationship to good gut health.10,11 5. Probiotics can help get mild diarrhea under control. Sometimes we just need a little help getting back to regular. Luckily, there have been many studies done on the relationship between probiotics and gastrointestinal comfort.12 Probiotics like Saccharomyces boulardii and the strain Bifidobacterium lactis HN019 have been studied extensively for their gastrointestinal benefits.13,14 6. Probiotics may help support mood & cognition. Our guts and brains communicate through what’s known as the gut-brain axis.15 In other words, just as stress or unhappiness can lead to an upset tummy, an upset microbiome can affect our mood.14 The opposite works as well: Recent studies have shown that probiotics may help support mood as well as cognition, even to the point of lowering stress levels.16,17 Curious which probiotics would be best for your specific needs? Your healthcare practitioner can help to guide you. For more information on nutrition and gut health, please visit the Metagenics blog. References: 1. Hill C et al. Natur Revs Gastro Hepatol. 2014;11(8):506—514. 2. Bermudez-Brito M et al. Ann Nutr Metab. 2012;61(2):160-174. 3. Kang E-J et al. Korean J Fam Med. 2013;34(1):2-10. 4. Reid G et al. FEMS Immunol Med Microbiol. 2001;32(1):37-41. 5. Reid G et al. FEMS Immunol Med Microbiol. 2006;30(1):49-52. 6. Reid G et al. J Med Food. 2004;7(2):223-228. 7. Reid G et al. FEMS Immunol Med Microbiol. 2003;35(2):131-134. 8. Loman T et al. BMC Public Health. 2013;13:259. 9. Stenman LK et al. EBioMedicine. 2016;13:190-200. 10. Ringel-Kulka T et al. J Clin Gastroenter. 2011;45:518-525. 11. Leyer GJ et al. Pediatrics. 2009;124:e172-179. 12. Vitetta L et al. Inflammopharmacol. 2014;22(3):135-154. 13. Kelesidis T. Therap Adv Gastroenterol. 2012;5(2):111–125. 14. Waller PA et al. Scand J Gastroenterol. 2011;46(9):1057–1064. 15. Carabotti M et al. Ann Gastroenterol. 2015:28(2):203-209. 16. Papalini S et al. Neurobiology of Stress. 2019;10:100141. 17. Akbari E et al. Front Aging Neurosci. 2016. https://www.frontiersin.org/articles/10.3389/fnagi.2016.00256/full. Accessed February 19, 2021. NCFM® and Bi-07® are registered trademarks licensed by DuPont. Submitted by the Metagenics team |
Categories
All
Archives
April 2024
|
|
Join Our Community
|
|
Amipro Disclaimer:
Certain persons, considered experts, may disagree with one or more of the foregoing statements, but the same are deemed, nevertheless, to be based on sound and reliable authority. No such statements shall be construed as a claim or representation as to Metagenics products, that they are offered for the diagnosis, cure, mitigation, treatment or prevention of any disease. |













 RSS Feed
RSS Feed