|
by Ashley Jordan Ferira, PhD, RDN
Recent research from three well-known cohorts, The Nurses’ Health Study (NHS), NHS2 and Health Professionals’ Follow-Up Study (HPFS), reveals that higher magnesium intake is associated with lower risk of type 2 diabetes (T2D), particularly in diets with poor carbohydrate quality.1 Green leafy vegetables, unrefined whole grains, and nuts are richest in magnesium, while meats and milk contain a moderate amount.2 Refined foods, like carbohydrates (carb), are poor sources of magnesium. Diets with poor carb quality are characterized by higher glycemic index (GI), higher glycemic load (GL), and lower fiber intake. These poor carbs require a higher insulin demand. The typical American diet is low in vegetables and whole grains, resulting in reduced magnesium intake. The Recommended Daily Allowance (RDA) for magnesium is 310-320 mg/day for adult women and 400-420 mg/day for adult men.3 Half of the US population fails to meet their daily magnesium needs, and hypomagnesemia exists in 1/3 of adults.4-5 Magnesium is needed for normal insulin signaling; current research has linked insufficient magnesium intake to prediabetes, insulin resistance and T2D.4 Increased magnesium intake has been inversely associated with T2D risk in observational studies.6 Collaborators from Tufts University, Harvard University, and Brigham and Women’s Hospital, sought to investigate the impact of magnesium intake, from both dietary and supplemental sources, on the risk of developing T2D in subjects who had diets with poor carb quality and raised GI, GL, or low fiber intake.1 They followed three large prospective cohorts, NHS, NHS2 and HPFS (totaling over 202,700 participants). Dietary intake was quantified by validated food frequency questionnaires (FFQ) every 4 years, and T2D cases were captured via questionnaires. Over 28 years of follow-up, there were 17,130 cases of T2D. Major study findings included:1
Similar to the US population estimates, 40-50% of study participants had inadequate magnesium intake. A healthful, varied diet and supplemental magnesium (especially in diets that restrict or exclude carbohydrates, dairy or meat) are essential to ensure sufficient daily magnesium intake. Why is this Clinically Relevant?
Citations
0 Comments
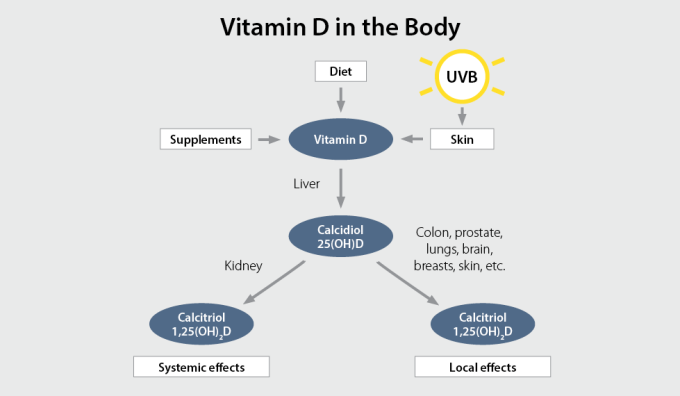
Did you know that getting just 10 minutes of sunshine (ultraviolet B, or UVB) per day helps the body create approximately 10,000 IU of vitamin D?1 This nutrient is necessary for the health of your bones, as well as overall health.2 However, during the months of November through February, and if you live north of Atlanta, there won’t be enough UVB rays to penetrate through the atmosphere and help your skin generate this vital nutrient. So is there something you can do? Sometimes you just have to create your own sunshine. And considering that three-quarters of teens and adults in the United States are deficient in vitamin D,3 as well as 1 billion people worldwide,4 this is where supplemental vitamin D can really help. Natural dietary sources of vitamin D are few (e.g. fatty fish, eggs), and fortified dietary sources such as milk, orange juice and cereal provide minimal amounts of vitamin D. This is why vitamin D is one of the most common nutrient gaps and also one of the easiest to address via supplementation. Why is vitamin D important? Vitamin D is a fat-soluble vitamin that regulates bone growth and mineralization and plays an important role in ensuring the muscles, heart, lungs, and brain function properly.2 Vitamin D has also been shown to support immune function. Vitamin D is not only an essential vitamin but also acts as a hormone in the body. Vitamin D that you obtain from the sun, food, beverage, or supplements must be first activated by the liver which converts the vitamin D to 25-hydroxyvitamin D (25(OH)D), also known as calcidiol.2 It is then converted by the kidneys and target tissues in the body to the biologically active form 1,25-dihydroxyvitamin D (1,25(OH)2D), also known as calcitriol.2 Calcitriol is the active, hormone form, which supports a variety of physiological functions, including helping the body regulate levels of calcium and phosphorus, as well as mineralize bone.5 Vitamin D deficiency What does it mean to be deficient in vitamin D? Measuring serum concentrations of 25(OH)D rather than 1,25(OH)2D is a better indicator of vitamin D status in the body due to its longer half life. Certain groups define vitamin D deficiency as 25(OH)D level less than 20 ng/mL (50 nmol/L).6 Vitamin D deficiency can be an issue for many people, including:7
Finding out if you need more vitamin D Measuring your vitamin D levels via a blood test is the only way to definitively know if you’re getting enough of this nutrient. With a 25(OH)D blood test from your healthcare practitioner, you will know your vitamin D levels and whether you need to take a supplement. Optimal levels of vitamin D vary according to different scientific organizations. For example, for adults the Vitamin D Council recommends daily supplementation with 5,000 IU of vitamin D3 when you cannot get enough sun to achieve a status between 40-60 ng/ml; whereas, the Endocrine Society recommends 1500-2000 IU/day.7-8 Higher levels are recommended to address deficiency.8 It is also important to recheck vitamin D levels two months after beginning a supplement regimen, and adjust as needed based on your practitioner’s recommendations. Which D is right for me? It’s important to get the form of vitamin D that is most bioavailable to the body. There are two kinds of vitamin D—D2 and D3. Vitamin D2 (ergocalciferol), is found in plants such as lichens and mushrooms, which are often irradiated by growers to boost nutritional value. Some soy and almond milks are also fortified with vitamin D2. Vitamin D3 (cholecalciferol) is the natural form of this nutrient that is created by the body with sun exposure, and research has shown that the D3 form increases the total circulating level of 25(OH)D significantly more effectively than D2.9-10 Vitamin D3 is found in small amounts in oily fish such as cod and salmon, egg yolks, as well as fortified cereals and milk, and some commercial mushrooms. Additionally, vitamin D3 has been shown to maintain adequate amounts of serum vitamin D levels during the winter months.11 There are also special sunlamps to help the skin generate vitamin D, but because of the risk for skin damage from ultraviolet rays, many healthcare practitioners don’t recommend using them. Make D your favourite letter for better health Whether you’re lucky enough to get the vitamin D you need from the sun all year around, or taking a vitamin D supplement, you’re wise to ensure you get enough of this vital nutrient. If you’re wondering whether you need more vitamin D, ask your healthcare practitioner. References:
Glyphosate, first registered for use in the United States in 1974, is an herbicide widely used to kill broadleaf weeds and grasses and regulate the growth of certain plants.1,2 It is employed in agriculture, forestry, in lawn and garden maintenance, and for weed control in industrial areas.1
The use of glyphosate has skyrocketed since 1997, after crops genetically modified to tolerate glyphosate were first introduced.3 The broad usage has led to questions of whether glyphosate remains present in the food we eat and water we drink, as well as whether it is safe for humans to consume. Because glyphosate binds so tightly to soil particles after use, it is believed to be prevented from entering groundwater.4 However, this may not be the case. During a 2002 study of waterways in nine Midwestern states, glyphosate was detected in 36% of the 154 samples taken, although the highest measured concentration was still well below the maximum contaminant level (set by the Environmental Protection Agency) of 700 micrograms per liter.3 And in one study of an agricultural community in Mexico, glyphosate was detected in both groundwater and bottled water.5 Due to its lack of quick degradation in plants, glyphosate residue could be present in the food supply.4 In tests performed by researchers and consumer watch groups, glyphosate has been detected in a number of foods, including bread, honey, oat-based cereals, granolas, and snack bars.6,7 Common means of glyphosate exposure Pure glyphosate is said to be low in toxicity, but it is often mixed with other ingredients that can make the resulting product more toxic.1 Directions for applying glyphosate usually caution users to wear gloves and eye protection to avoid skin and eye irritation and to be careful not to breathe the compound to protect against nose and throat irritation.1 Although it isn’t easy for glyphosate to pass through the skin into the body, it is possible to ingest glyphosate by breathing it in while spraying or by eating or smoking after applying, without first washing your hands.1 When absorbed or ingested, most glyphosate tends to remain unchanged and leaves the body pretty quickly in urine or excrement.1 Despite that, swallowing glyphosate-containing products can result in nausea, vomiting, diarrhea, and burns in the mouth and throat.1 Intentional ingestion has resulted in fatalities in some cases.1 Because glyphosate products often contain additional chemicals, it can be hard to determine whether glyphosate alone is responsible for the adverse symptoms observed when humans come into contact with it.8 For instance, some studies suggest that the surfactant polyoxyethyleneamine used with glyphosate is more toxic than glyphosate alone.8 Agency opinions and glyphosate research Consensus on risks associated with glyphosate exposure has been tough to pin down. In April 2019, the US Environmental Protection Agency stated it “continues to find [glyphosate] poses no risk to public health when used as labeled and that it is not a carcinogen.”2 This follows a similar declaration by the European Food Safety Authority in 2015 that glyphosate is unlikely to be a carcinogen in humans.9 By contrast, the World Health Organization’s International Agency for Research on Cancer (IARC) stated in March 2015 that it had analyzed the results of over 1,000 studies and determined that glyphosate was possibly carcinogenic to humans.10 In an updated monograph released one year later, the IARC attributed the difference between its statement and the findings of other agencies to the source material, noting that most regulatory agencies reviewed nonpublic industry data from toxicological studies.10 Still, research into glyphosate’s possible carcinogenic effects continues. A recent analysis of human epidemiological studies confirmed a link between glyphosate exposure and an increased risk for non-Hodgkin’s lymphoma.11 And in another lab study, glyphosate was shown to stimulate the growth of a certain type of hormone-dependent breast cancer in cells by acting on estrogen receptors, indicating it may be an endocrine disruptor.12 Researchers are also investigating glyphosate’s impact on the human reproductive system. In one study, exposure to glyphosate-based herbicides below the toxicity threshold decreased the activity of aromatase, a key enzyme in balancing sex hormones.13 Another study in prepubertal male rats showed a decrease in testosterone levels in those given soy milk supplemented with glyphosate.14 And results of a study in pregnant mice indicate that glyphosate can cause the ovaries to fail and interfere with secretion of hormones.15 There have been cases of accidental exposure to concentrated glyphosate solutions causing neurological lesions, suggesting glyphosate could be neurotoxic in high doses.16 In a model of the blood-brain barrier, researchers observed that a high dose of glyphosate resulted in neurological damage and altered metabolism of glucose.16 Glyphosate and antibiotic resistance Recent research has uncovered a worrisome link between glyphosate use and the growing problem of antibiotic resistance.17,18 It is believed the use of glyphosate is leading to changes in microbiome composition and, as a result, increases in resistance to critical antibiotics.17 Researchers note that when bacteria are exposed to non-antibiotic chemicals such as herbicides, they can be inclined to develop resistance to antibiotics more rapidly: in some cases, 100,000 times faster.18 Looking to avoid glyphosate? The Detox Project offers Glyphosate Residue Free and Gold Standard Detox certification to bring a new era of transparency to the food and supplement industries. References:
By Michael Stanclift, ND You’re feeling that rush as you catch your stride on your morning run. The air is perfect. Suddenly a cramp or muscle ache stops you dead in your tracks. You try to shake it off, but it just grabs more. Ah! We still don’t completely understand why muscles tighten up involuntarily. Exercise, pregnancy, electrolyte imbalances, nerve compression, and diminished blood supply to the muscle all may contribute.1,2 So what can we do to combat these harmless but pesky discomforts? In this article we’ll look at what the research says. Surprisingly, some popular natural remedies don’t shine through in the current medical evidence. What might not help with cramping Magnesium and Epsom salts: A Cochrane Review found that oral magnesium wasn’t likely to help with muscle cramps in older people, and the findings were inconsistent in pregnant women.3 A recent randomized, placebo-controlled trial in pregnant women found no difference in leg cramps with magnesium compared to placebo.4 Epsom salt (magnesium sulfate) baths have long been a go-to for muscle relaxation, and “float” centers with sensory deprivation tanks full of the magnesium-rich water have popped up as an urban refuge from the constant stimulation of modern life. A study in nonathletic healthy men found a one-hour float (in magnesium sulfate) after exercise reduced pain perception compared to one hour of passive recovery.5 However, these findings are tough to attribute to magnesium, as the study’s control didn’t match other potentially therapeutic factors, such as body positioning and sensory deprivation.5 So a relaxing bath may help with cramping and muscles, but it’s unclear if adding Epsom salt makes a significant difference. Active cool-down and static stretching: Many believe after exercising intensely a period of low-to-moderate intensity will prevent muscle soreness and injuries, but this doesn’t appear to be true.6 A 2018 review found evidence on active cool-downs shows it doesn’t significantly reduce soreness, stiffness, or range of motion and may inhibit muscular glycogen resynthesis (energy storage).6 This same review found that static stretching before or after exercise didn’t reduce muscle soreness.6 What might help with muscle cramping: Foam rolling: This surprisingly simple tool can be valuable if you suffer from muscle soreness and cramps. Using a foam roller after exercise can reduce muscle soreness and improve athletic performance the following day.6 Physical therapists from Harvard agree that 30-120 seconds per area can be helpful in relieving sore muscles and preventing cramps.7 Muscle soreness: Tart cherry or pomegranate juice: A small randomized, double-blind, placebo-controlled trial found 355 ml (~12 oz.) of tart cherry juice drunk twice a day for a week before a 26 km (16-mile) run reduced the amount of pain reported from participants.8 A research review found similar effects from drinking tart cherry juice twice a day, and one study found pomegranate juice reduced soreness.9 But the research on these two drinks in relation to muscle soreness has shown mixed results.9 A recent study compared tart cherry, pomegranate, and placebo drinks to analyze the impact on muscle soreness in nonresistance trained men.10 In this study, the researchers were surprised to find that neither of the fruit drinks appeared to help with muscle soreness when compared to placebo.10 Ginger: In a small double-blind, randomized, placebo-controlled trial in experienced runners, 5 days of powdered ginger supplementation (1.4 g/day) moderately reduced muscle soreness from a run (on day 3) during the supplement period.11 A review of randomized clinical trials found that consuming up to 4 g of ginger postintense exercise can reduce muscle soreness and improve muscle recovery.12 Lower single dosages of 2 g ginger did not help with muscle soreness when compared to placebo.12 This suggests it may take multiple days or higher doses to get the effect. Curcumin: It’s no surprise that curcumin, a bright orange compound from the spice turmeric is making news again. A research review found curcumin in a wide range of doses (150 mg-5,000 mg) can reduce muscle soreness after exercise.13 Curcumin can work when used on an “as needed” basis, with even a single dose (150-200 mg) showing effectiveness for muscle soreness following exercise.13 Interestingly, in this review they found small doses (90 mg twice a day) of curcumin taken for 7 days before exercise had no effect on postexercise soreness, while the same dosage taken after exercise for 4 days was effective.13 Other studies in the review at similar doses did not find curcumin improved muscle soreness compared to placebo, so differences in the trial participants and types of exercise may influence the effects.13 Conclusion:Cramping and muscle soreness can ruin a good exercise session, but they don’t have to. When it comes to combatting these annoying aches, you have numerous options—but beware that some popular natural treatments might be more hype than help.
References: 1. Young G. Leg cramps. BMJ Clin Evid. 2015;2015:1113. 2. Mayo Clinic Staff. Muscle cramps. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/muscle-cramp/symptoms-causes/syc-20350820#:~:text=Overuse%20of%20a%20muscle%2C%20dehydration,Inadequate%20blood%20supply. Accessed February 11, 2021. 3. Garrison SR et al. Magnesium for skeletal muscle cramps. Cochrane Database Syst Rev. 2012;2012(9):CD009402. 4. Araújo CAL et al. Oral magnesium supplementation for leg cramps in pregnancy-An observational controlled trial. PLoS One. 2020;15(1):e0227497. 5. Morgan PM et al. The acute effects of flotation restricted environmental stimulation technique on recovery from maximal eccentric exercise. J Strength Cond Res. 2013;27(12):3467-3474. 6. Van Hooren B et al. Do we need a cool-down after exercise? A narrative review of the psychophysiological effects and the effects on performance, injuries and the long-term adaptive response. Sports Med. 2018;48(7):1575-1595. 7. Harvard Health Staff. Roll away muscle pain. Harvard Health Publishing. https://www.health.harvard.edu/staying-healthy/roll-away-muscle-pain#:~:text=Foam%20rollers%20are%20easy%2Dto,from%20exercise%2C%20and%20reduce%20injury.&text=As%20you%20age%2C%20occasional%20muscle,lightweight%20cylinder%20of%20compressed%20foam. Accessed February 11, 2021. 8. Kuehl KS et al. Efficacy of tart cherry juice in reducing muscle pain during running: a randomized controlled trial. J Int Soc Sports Nutr. 2010;7:17. 9. Bowtell J et al. Fruit-derived polyphenol supplementation for athlete recovery and performance. Sports Med. 2019;49(Suppl 1):3-23. 10. Lamb KL et al. No effect of tart cherry juice or pomegranate juice on recovery from exercise-induced muscle damage in non-resistance trained men. Nutrients. 2019;11(7):1593. 11. Wilson PB. A randomized double-blind trial of ginger root for reducing muscle soreness and improving physical performance recovery among experienced recreational distance runners. J Diet Suppl. 2020;17(2):121-132. 12. Rondanelli M et al. Clinical trials on pain lowering effect of ginger: A narrative review. Phytother Res. 2020;34(11):2843-2856. 13. Yoon WY et al. Curcumin supplementation and delayed onset muscle soreness (DOMS): effects, mechanisms, and practical considerations. Phys Act Nutr. 2020;24(3):39-43. By Monazza Ahmad, B.Pharm, MSc
A healthy genital tract makes some of the most important phases in a woman’s life more enjoyable and easier to manage. Menstruation, intimacy, giving birth, and embracing menopause are all significant milestones. But you may not realize that good vaginal health plays a fundamental role in reaching these milestones with minimum hurdles. Importance of gut health is well-researched and well-understood for our overall health, concluding that the gut is vital to the health of most organs in our body. Gut health is defined by the microbiome that resides in it and influences the absorption and digestion of everything we consume. These microorganisms constantly and selectively translocate to different parts of the body, creating a unique microecosystem in each organ.1 Curious how the vaginal microbiome differentiates from the gut microbiome? To understand this concept better, we will briefly explore the diversity of the microbiome in the human body. Diversity of human microbiomeMicrobiome diversity in our body depends on factors like diet, environment, genetics, and early exposure to microbiota, meaning at birth. Just like skin and scalp, the female genital tract is also represented by its own community of microorganisms. The vaginal microbiota is evolved through a continuous translocation of species from gut to vagina or from a mother to child at birth.1 Driven by hormonal changes, the complex vaginal microbiome is continuously transformed throughout various cycles of the female lifetime—from birth to puberty, menstruation, pregnancy, menopause, and postmenopause.1 Difference between gut and vaginal microbiomeSimply put, gut microbiota is more diverse while vaginal microbiota is more selective in healthy bacterial strains. This means using the same interventions to protect vaginal flora as that for the gut may not always be optimal. Recognizing the difference between the native microbiome of gut and vagina helps us find the right and safe solutions to help avoid undesirable genital conditions.1 It is important to understand that the physiological and biochemical characteristics of microorganisms remain the same in the gut and vagina; however, their immune responses vary considerably between the two environments.1 In other words, the way each environment detects a substance as harmful or safe is different.1 For example, where the by-products of bacterial fermentation (such as short-chain fatty acids) have shown to prevent damage to the gastrointestinal tract, they have shown unfavorable effects in the genital tract, leading to negative outcomes in the reproductive and gynecological system.1 When the undesirable bacteria from the gut invade the vaginal or urethral area, they create an imbalance that may lead to various female concerns. What is the gut-vagina axis?Modern research has revealed a collection of intricate pathways, namely gut-vagina axis, which connects the vaginal tract to our gut. Despite the difference between intestinal flora and the vaginal flora, the former is found to be involved in the development of some vaginal imbalances. For example, bacteria from the gut pass to the rectum, from where they can travel to the vagina due to the close proximity, resulting in unfavorable conditions. Therefore, it is important to keep the communication smooth between the gut and vagina.2 Effects of vaginal microbial imbalanceVaginal microbial imbalances may result in several unfavorable conditions.
Factors affecting the vaginal microbiomeThe intricate and dynamic female genital tract requires special attention to care. Here are some of the factors that can create imbalance in the vaginal microbiome.
References: 1. Amabebe E et al. Front Immunol. 2020;11:2184. 2. Brannon JR et al. Nat Commun. 2020;11:2803. 3. Han Y et al. Front Micriobiol. 2021;12:643422. 4. Lehtoranta L et al. Front Micriobiol. 2022;13:819958. 5. Gholiof M et al. Front Reprod Health. 2022;4:963752. 6. Lewis FMT et al. Obstet Gynecol. 2017;129(4):643–654. 7. Cheng G et al. Eukaryot Cell. 2006;5(1):180–191. 8. Mirmonsef P et al. PLoS One. 2014;9(7):e102467. 9. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/yeast-infection/symptoms-causes/syc-20378999. Accessed December 5, 2022. 10. Gupta K et al. J Infect Dis. 2000;181(2):595-601. 11. Szymański JK et al. Int J Environ Res Public Health. 2021;18(9):4935. 12. Neggers YH et al. J Nutr. 2007;137(9):2128-2133. 13. Tohill BC et al. Am J Clin Nutr. 2007;85(5):1327-1334. Every BODY and every age have unique nutritional needs. However, with every stage of life, there are common nutritional inadequacies that women are most likely to experience. Learn what supplements are best for YOU! 20s You may find yourself strapped for time and cash during these exciting transitional years, which could result in an unbalanced diet. What supplements should you consider taking?
In your 30s, you may be expanding your family and/or career, so you need all the energy you can get!
Your 40s are filled with possibilities; whether you are starting a new business, climbing the corporate ladder, juggling older kids, or welcoming a new addition, women in their 40s are thriving!
Your 50s and beyond can be a great time to reconnect with yourself and maybe your spouse too. Make sure to support all the amazing years ahead of you with high-quality supplements.
References
By Monazza Ahmad, B.Pharm, MSc
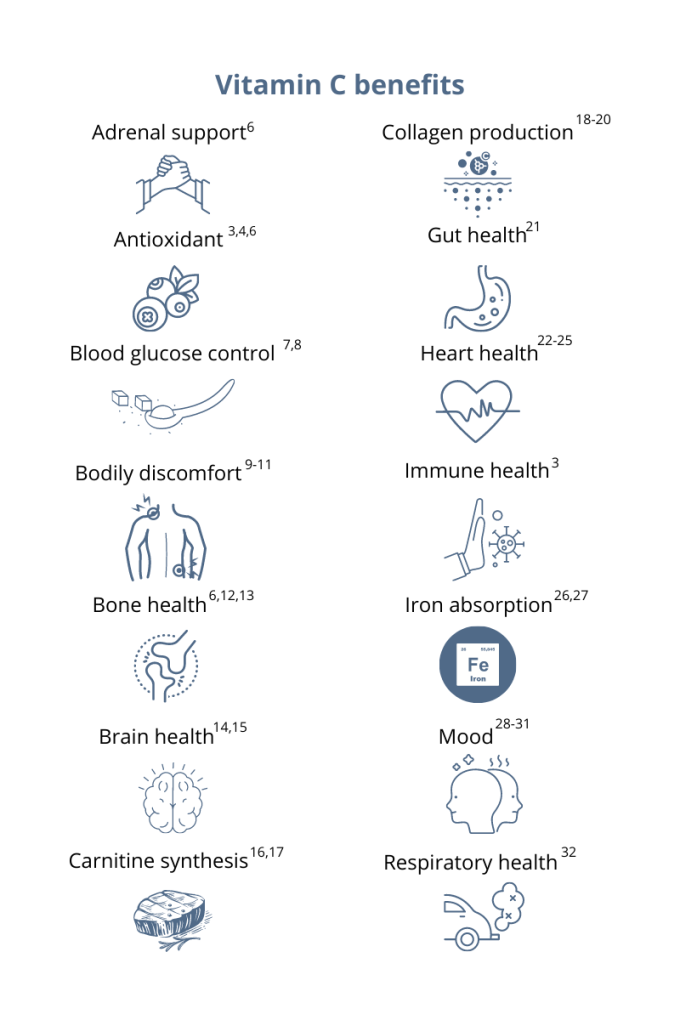
One of the symptoms of menopause that women wish had a permanent cure is hot flashes! The most uncomfortable and rather embarrassing aspect of hot flashes is that they visit unexpectedly. It doesn’t matter if you’re in a work meeting with executives, teaching to kindergarteners, at an intimate dinner, or just by yourself, there is no time and place for its occurrence.1 The sudden attack of heat and an outburst of sweating is enough to make anyone nervous. Prescription treatment options are usually costly and not suitable for some health conditions,2,3 leaving many women with no choice but to ignore it despite the distressful effect hot flashes have on their quality of life.4,5 But don’t be disappointed. In this post, you will find various natural ways and nonhormonal options to find relief from hot flashes until they disappear after completing their course. The severity and duration vary from woman to woman, so let the treatment take its time.5 Let’s understand what’s going on behind the scenes with hot flashes so we can mitigate the chaos it creates based on our body’s bioindividual needs. How do I know it’s a hot flash? During a hot flash, there is a sudden burst of heat from chest up that may result in red blotches on your skin and uncontrollable sweat. The untimely sweating, flushing, heat, and chills a few times a week or several times in a day is normal during menopause but definitely frustrating. A single episode of hot flashes can last anywhere from one to five minutes and up to an hour. The hot flashes range anywhere from 1 to 10 daily with the duration from a few years to a couple of decades.5 What’s causing this gush of sweat? Research suggests that hot flashes are a possible result of hypothalamus activity in regulating body’s temperature due to estrogen fluctuation. Here’s what that means: Reduced estrogen levels can cause blood vessels near the skin to dilate, increasing the blood flow that elevates body temperature. This increase in temperature triggers hypothalamus (your body’s thermostat) to detect warmth so it starts to cool down the body by a process called sweating.4,5 We all know that sweat is an important mechanism of temperature control in the body. It helps dissipate body heat by turning heat vapors into liquid (sweat) to keep the body cool. So you really don’t want to stop a hot flash, since it’s protecting our bodies from getting overheated. What is needed is hormone regulation so the hot flashes are less severe. How to squash hot flashes While hot flashes subside on their own and may not require treatment, those experiencing severe effects may want to seek safe relief options. Here are some lifestyle improvements and natural remedies that might be helpful in tackling hot flashes safely. Make sure to consult with your healthcare practitioner before taking any action; they will advise you on the best options and the right dosage for your specific health needs. NUTRITIONAL SUPPORT Vitamin D: Reduced levels of vitamin D are associated with many health conditions, including hot flashes. Try getting at least 15 minutes of sun every day and eat vitamin D-rich foods.6,7 Vitamin E and curcumin: Both these supplements have shown to reduce the severity and number of hot flashes. The dose of 200 to 400 IU for vitamin E and 500 mg for curcumin is found to be effective.8,9 Give at least four weeks to curcumin and up to eight weeks to vitamin E to work.3 Folic acid: About 1 mg of folic acid, a form of vitamin B, has shown weekly gradual improvement in severity. It may take about 3-4 weeks to reduce frequency and the duration of hot flashes.10 Vitamin B6 and oily fish: The impact of this vitamin on brain health, immune system, nerve function, and the breakdown of macronutrients explains its positive effect on vasomotor symptoms of menopause.11,12 To avoid pills, taking this vitamin in the form of oily fish has shown great results, too.13 ALTERNATIVE APPROACHES Plant-derived solutions: Black cohosh,14,15 phytoestrogens, and some other herbs are commonly used for hot-flashes relief.16 Read more about these options on our blog: “Consider Using Plant-Derived Solutions to Treat Menopausal Symptoms.” ERr 731®:* An extract of Siberian rhubarb, is the nonhormonal and generally safe solution16 to have shown significant improvements in all 11 major menopausal symptoms.17* It also has a high recommendation rate not only from providers but also from women who use it. Acupuncture: This form of complementary therapy, in addition to usual care, has shown marked improvement in hot flashes and other menopausal symptoms. Acupuncture is commonly used for stress management, pain relief, and overall wellness with neurological stimulation.3,18,19 LIFESTYLE FACTORS Resistance training: Lifting safe weights and isometric and plyometric exercises helps reduce hot flashes. In a study of menopausal women, hot flashes reduced by more than 40% in less than four months in women who performed these exercises regularly for at least three times per week. Active women experience less severe symptoms than inactive women.20,21 Eating: Food intake at regular intervals can reduce hot-flash frequency. So have a healthy snack between meals to avoid letting your blood glucose levels fall too low.22 Diet: A low-fat, plant-based diet including half cup of cooked soybeans 3-4 times daily along with TLC diet can significantly reduce the frequency and severity of hot flashes and other menopausal symptoms.23,24 Stress management: Anxiety and stress are the common triggers for many health conditions. Stress reduction techniques such as meditation, yoga, deep breathing, and resting can all help with hot flashes.25 Also of vital importance: Avoiding alcohol, smoking, spicy food, caffeine, and sugar, along with physical exertion and managing weight all contribute toward reducing the severity of hot flashes.26,27 Menopause and its symptoms are unavoidable, but you can definitely take these steps to minimize the discomfort and enjoy your daily activities. A piece of advice for younger women and those who haven’t reached menopause yet: Start taking care of your body early on in life. Eat healthy food and do regular physical activity to keep your hormones regulated. So when menopause hits, your body will be prepared to handle the ups and downs of aging and can recover faster. References: 1. Mayo Clinic. Hot flashes: symptoms & causes. https://www.mayoclinic.org/diseases-conditions/hot-flashes/symptoms-causes/syc-20352790. Accessed February 14, 2023. 2. Rosenberg V et al. Hormone-replacement therapy and its association with breast cancer subtypes: a large retrospective cohort study. Int. J Womens Health. 2021;1:1207-1216. 3. Mayo Clinic. Hot flashes: diagnosis & treatment. https://www.mayoclinic.org/diseases-conditions/hot-flashes/diagnosis-treatment/drc-20352795. Accessed February 14, 2023. 4. Bansal R et al. Menopausal hot flashes: a concise review. J Midlife Health. 2019;10(1):6-13. 5. Freedman RR. Menopausal hot flashes: mechanisms, endocrinology, treatment. J Steroid Biochem Mol Biol. 2014;142:115-120. 6. Arslanca T et al. The relationship between vitamin D and vasomotor symptoms during the postmenopausal period. Clin Lab. 2020;66(7). 7. Religi A et al. Estimation of exposure durations for vitamin D production and sunburn risk in Switzerland. J Expo Sci Environ Epidemiol. 2019;29(6):742-752. 8. Ataei-Almanghadim K et al. The effect of oral capsule of curcumin and vitamin E on the hot flashes and anxiety in postmenopausal women: a triple blind randomised controlled trial. Complement Ther Med. 2020;48:102267. 9. Ziaei S et al. The effect of vitamin E on hot flashes in menopausal women. Gynecol Obstet Invest. 2007;64(4):204-207. 10. Bani S et al. The effect of folic acid on menopausal hot flashes: a randomized clinical trial. J Caring Sci. 2013;2(2):131-140. 11. Harvard T.H. Chan School of Public Health. Vitamin B6. https://www.hsph.harvard.edu/nutritionsource/vitamin-b6. Accessed February 14, 2023. 12. National Institutes of Health. Vitamin B6 fact sheet for consumers. https://ods.od.nih.gov/factsheets/VitaminB6-Consumer. Accessed February 14, 2023. 13. Odai T et al. Severity of hot flushes is inversely associated with dietary intake of vitamin B6 and oily fish. Climacteric. 2019;22(6):617-621. 14. National Institutes of Health. Black cohosh. https://www.nccih.nih.gov/health/black-cohosh. Accessed February 14, 2023. 15. Geller SE et al. Botanical and dietary supplements for menopausal symptoms: what works, what does not. J Womens Health (Larchmt). 2005;14(7):6340649. 16. Chang JL et al. Rheum rhaponticum extract (ERr 731): postmarketing data on safety surveillance and consumer complaints. Integr Med (Encinitas). 2016;15(3):34-39. 17. Kaszkin-Bettag M et al. Confirmation of the efficacy of ERr 731 in perimenopausal women with menopausal symptoms. Altern Ther Health Med. 2009;15(1):24-34. 18. Kim KH et al. Effects of acupuncture on hot flashes in perimenopausal and postmenopausal women–a multicenter randomized clinical trial. Menopause. 2010;17(2):269-280. 19. Mayo Clinic. Acupuncture. https://www.mayoclinic.org/tests-procedures/acupuncture/about/pac-20392763. Accessed February 14, 2023. 20. Berin E et al. Resistance training for hot flushes in postmenopausal women: A randomised controlled trial. Maturitas. 2019;126:55-60. 21. Dabrowska-Galas M et al. High physical activity level may reduce menopausal symptoms. Medicina (Kaunas). 2019;55(8):466. 22. Dormire S et al. The effect of dietary intake on hot flashes in menopausal women. J Obstet Gynecol Neonatal Nurs. 2007;36(3):255–262. 23. Barnard ND et al. The Women’s Study for the Alleviation of Vasomotor Symptoms (WAVS): a randomized, controlled trial of a plant-based diet and whole soybeans for postmenopausal women. Menopause. 2021;28(10):1150-1156. 24. Welty FK et al. The association between soy nut consumption and decreased menopausal symptoms. J Womens Health (Larchmt). 2007;16(3):361-369. 25. Freeman EW et al. Anxiety as a risk factor for menopausal hot flashes: evidence from the Penn ovarian aging cohort. Menopause. 2016;23(9):942–949. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4993654/ 26. NIH. Hot flashes: what can I do? https://www.nia.nih.gov/health/hot-flashes-what-can-i-do. Accessed February 14, 2023. 27. Herber-Gast GCM et al. Fruit, Mediterranean-style, and high-fat and -sugar diets are associated with the risk of night sweats and hot flushes in midlife: results from a prospective cohort study. Am J Clin Nutr. 2013;97(5):1092-1099. You’ve probably heard that vitamin C supports your immune system. This essential micronutrient seems to be everywhere! And it’s a good thing because, unlike most mammals, humans can’t synthesize vitamin C on their own.1 Also, vitamin C is water-soluble, which means the body quickly loses this essential vitamin through urine, so it’s important to make vitamin C a daily part of your diet.1 Having extremely low levels of vitamin C for prolonged periods can result in scurvy, a historical disease linked to pirates and sailors who faced long journeys at sea without fresh fruits and vegetables. While cases of scurvy in the United States are rare, a recent study reported that 31% of the US population are not meeting the daily recommended intake of vitamin C.1 Greater than 6% of the US population are severely vitamin C deficient, while low levels of vitamin C, associated with weakness and fatigue, were observed in 16% of Americans.2 As a whole, 20% of the US population showed marginally low levels of this essential micronutrient.2 How much vitamin C do I need?The US recommended daily dietary allowance of vitamin C is 75 mg for women and 90 mg for men.3 Experts recommend an estimated 200 mg of vitamin C daily for favorable health benefits.4 Adults can take up to 2,000 mg of vitamin C per day; however, high doses of vitamin C may cause diarrhea, nausea, and stomach cramps.5 Due to the varying health needs of individuals, it’s always a good idea to work with your healthcare practitioner to ensure that you are getting the right amounts of micronutrients in your daily diet. Where can you find this marvelous, multifaceted micronutrient? Ready to add vitamin C to your daily regimen? Talk to your healthcare practitioner about how much would be right for you.
References: 1. Granger M et al. Adv Food Nutr Res. 2018;83:281-310. 2. Schleicher RL et al. Am J Clin Nutr. 2009;90(5):1252-1263. 3. https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/. Accessed August 3, 2021. 4. Frei B et al. Crit Rev Food Sci Nutr. 2021;52(9):815-829. 5. Hathcock JN et al. AM J Clin Nutr. 2005;81(4)736-745. 6. Ashor AW et al. Eur J Clin Nutr. 2017;71(12):1371-1380. 7. Mason SA et al. Free Radic Biol Med. 2016;93:227-238. 8. Chen S et al. Clin J Pain. 2016;32(2):179-185. 9. Carr AC et al. J Transl Med. 2017;15(1):77. 10. Dionne CE et al. Pain. 2016; 157(11):2527-2535. 11. Chin KY et al. Curr Drug Targets. 2018;19(5):439-450. 12. Ratajczak AE et al. Nutrients. 2020;12(8):2263. 13. Dixit S et al. ACS Chem Neurosci. 2015;6(4):570-581. 14. Monacelli F et al. Nutrients. 2017;9(7):670. 15. Johnston CS et al. J of Nutr. 2007;137(7):1757–1762. 16. Johnston CS et al. Nutr Metab (Lond). 2006;3(35):1743-7075. 17. Moores J. Br J Community Nurs. 2013;Suppl:S6-S11. 18. Carr AC et al. Nutrients. 2017;9:1211. 19. Shaw G et al. Am J Clin Nutr. 2017;105(1):136-143. 20. Ratajczak AE et al. Nutrients. 2020;12(8):2263. 21. Ashor AW et al. Nutr Res. 2019;61:1-12. 22. Moser MA et al. Int J Mol Sci. 2016;17(8):1328. 23. Wu JR et al. J Cardiovasc Nurs. 2019;34(1):29-35. 24. Akolkar G et al. Am J Physiol Heart Circ Physiol. 2017;313(4):H795-H809. 25. Cook JD et al. Amer J Clin Nutr. 2001;73(1):93-98. 26. Saunders AV et al. Med J Aust. 2013;199(S4):S11-S16. 27. Amr M et al. Nutr J. 2013;12:31. 28. Consoli DC et al. J of neurochem. 2021;157(3):656-665. 29. Bajpai A et al. J Clin Diagn Res. 2014;8(12):CC04-CC7. 30. Koizumi M et al. Nutr Res. 2016;36(12):1379-1391. 31. Whyand T et al. Respir Res. 2018;19(1):79. 32. Azuma A et al. Tairyoku Kagaku Japanese J of Phys Fit and Sports Med. 2019;68(2):153-157. By Melissa Blake, ND
Our bodies have an amazing natural ability of keeping to a daily schedule via an internal 24-hour master clock.1 This clock contributes to the patterns, also known as circadian rhythms, of many biological activities including sleep-wake cycles, eating patterns, and hormone function.1 Finding ways to support and balance this clock, along with the many systems it regulates, may offer a novel way to optimize health. One way to optimize is through diet timing. The circadian diet as a way of eating takes into account not only what we eat, but when.2 It is an approach to eating that synchronizes food intake around our biological clocks, emphasizing eating in sync with the body’s natural tendencies and instincts. This means eating during daylight hours or hours when we are naturally more active. Eating in this way can help support circadian rhythms and contribute to overall health and wellness.2 As diets and terms including intermittent fasting, circadian diet, and time-restricted eating gain popularity, the question may arise as to whether the principle on which this “circadian approach” applies to other aspects of nutrition, including supplementation. 5 common supplements Although there’s still much to learn about optimal timing for both food and supplements, current evidence suggests it may play a role.3 Here are a few general guidelines for five common supplements to help you add the extra layer of timing and optimize your plan: B complex B vitamins are often recommended to support healthy energy and mood.4 There is some evidence that taking B vitamins before bed can have a negative effect on sleep quality.5 Consider taking any B vitamins, including a B complex, earlier in the day with food. Fish oil The most common complaints I hear about fish oil are burping or nausea. Taking fish oil supplements with food, divided into two doses, may help reduce these harmless yet annoying side effects. Magnesium Magnesium is an essential micronutrient that plays a role in hundreds of reactions in the body.6 Due to the overall benefits of magnesium supplementation, consistency is more important than any timing in this case. Known for muscle relaxation and improved sleep, taking magnesium before bed may enhance those benefits in some people.7 Others may notice digestive issues and may choose to take with food. Probiotics Any recommendation related to probiotic supplementation should be based on the specific strain; however, much of this detailed evidence does not yet exist. Meal timing has more or less of an impact on probiotics depending on the strain, the dose, delivery method, etc.8 The consensus, however, is to take probiotics 30 minutes before or during a meal versus after eating.9 Another guideline is to space probiotics away from antibiotic medications by two hours to reduce interaction. Vitamin D Along with vitamins A, E, and K, vitamin D is a fat-soluble vitamin that is better absorbed when taken with a fat-containing meal (ex. fatty fish, avocado, olive oil, cheese, eggs).10 Although we do not have substantial evidence to support specific timing, it may make sense to take vitamin D supplements in the morning with breakfast to mimic the timing of exposure to natural sunlight. Summary As we continue to learn about specific supplements and optimal timing, consider that the best timing is the one you can stick with. You cannot benefit from a supplement you do not take. The most important thing is to take your supplements at a time that is convenient for you so you can be consistent. Work with a knowledgeable healthcare provider to determine the optimal plan for you that includes quality, quantity, and timing. References:
By Cassie Story, RDN
We are social creatures. Hardwired in our being is the desire to belong, fit in, and play with our fellow humans. In fact, survival depends on our ability to bond. So what happens to your mental state when you remove yourself from others over the course of a year and a half? While long-term data on the mental health implications of isolation are currently being studied, we do have short-term studies emerging, which evaluated mental health concerns over the past year. A new phenomenon has occurred for workers who have been utilizing online video platforms for a majority of their day-to-day work. “Zoom fatigue” may have inadvertently activated the fight or flight system within the brain.1 This is according to a recent report from Stanford that offers four potential aspects of Zoom fatigue and the unintended psychological consequences that follow.1 With this knowledge, it is important to recognize the spectrum of emotions that you might be feeling as you prepare to “get back” into the real world—to step out from behind the screen and live life again. This article identifies common emotions that you may be experiencing, effective mantras for change, and supportive nutrients that can play a role in taking you from feeling blah to rah! Emotional state: fear & anxietyIt is understandable to have some lingering fear and anxiety about social reentry. Due to our constant connection to news and information, many people have likely experienced some level of fear and anxiety that is atypical for themselves over the past year. It wouldn’t be realistic to expect yourself to go from living in that state of mind, to complete joy and jubilation. Mantra: I am safe Supportive nutrients: palmitoylethanolamide (PEA) and hemp PEA is a bioactive lipid molecule that the body produces naturally and is found in the lipid extracts of foods.2 PEA has been researched for over 70 years and has a wide range of clinical applications from mild bodily discomfort and immune system support to mood and neurological health.2-6 Full-spectrum hemp is sourced from aerial plant parts including the stalk, stems, seed, and flower of the hemp plant and contains beneficial phytocannabinoids and terpenes. They work together to produce a synergistic effect on the endocannabinoid system, which has been found to support a healthy stress response and has positive neurological benefits.7 Emotional state: stressLoneliness and isolation have been found to increase stress levels in the body.8 Couple that with uncertainty about the future and worry for your own health and wellbeing, as well as that of your friends and family, and this creates a perfect brew for stress to thrive. Are you ready to reclaim your balance? Mantra: I am at peace Supportive nutrient: phosphatidylserine Phosphatidylserine is an important phospholipid in the brain and a key building block of nerve cell membranes.9 Research suggests that it may support mental focus and help support a healthy endocrine response to acute mental stress.10 Emotional state: low self-confidence or decline in body imageFor most people, due in part, to our society’s environmental factors of convenience foods and little built-in day-to-day movement, maintaining health and wellbeing requires routine and planning. Removing ourselves from typical day-to-day activities, and increasing a sedentary lifestyle, may lead to undesired weight gain. If you have experienced an increase in body weight, first remind yourself, you are not alone. Second, find something about your body to be grateful for right now. Third, speak kindly to yourself. If you have now found that a majority of your “real life” clothes no longer fit, maybe it is time to implement a supportive weight-loss routine. Mantra: I am strong (or flexible, sexy, healthy) Supportive nutrient: meal replacements Meal replacements are reduced-calorie portion-controlled products often fortified with micronutrients. Studies show that replacing just one or two meals per day with a meal-replacement product produces greater total weight loss, and a greater proportion of participants meet their total weight-loss goals both in the short- and long-term, compared to a low-calorie diet without the use of meal replacements.11 Emotional state: lack of motivationSpending the majority of your time in a seated position, whether the couch or an office chair, can wreak havoc on motivation levels. Going from staring at one screen to the next, between your computer, TV, and phone, can cause a numbness of sorts and lead to lack of desire or motivation to try new things. We are hardwired to want to try new things. Being limited from participating in your favorite activities may have decreased your motivation without your realizing it. Mantra: I enjoy new experiences Supportive nutrients: holy basil, ashwagandha, amla fruit These are a classic blend of Ayurvedic herbs. Holy basil (Ociumum sanctum) leaves and stems contain a variety of compounds including triterpenes (oleanolic and ursolic acid), saponins, flavonoids, and phenols.12 Roots of ashwagandha (Withania somniferum), an herb grown in India, contain withanolides.13 Amla fruit, or Indian gooseberry (Phyllanthus emblica), is rich in vitamin C, a potent antioxidant.14 Research suggests these adaptogens may support the body in adjusting to various stressful environmental challenges.15 These herbs may help reduce some of the challenges associated with stress.16 Emotional state: excitementPerhaps you are experiencing a different type of emotion than those listed above. Maybe you are excited and “champing at the bit” to get back out into your typical social life. If you feel that you need some calming support because you just cannot wait to get back out there, here are some things to consider: Mantra: I am calm Supportive nutrients: folate, magnesium, vitamins B12 and B6 Folate & vitamin B12 are cofactors in the synthesis of neurotransmitters, including serotonin.17-18 Serotonin is associated with mood, sleep, and relaxation.19 Vitamin B6 is a factor in the body’s conversion of glutamate, which is a stimulatory neurotransmitter, into gamma-amino-butyric acid (GABA), which is associated with calming and relaxation.20 Magnesium is an essential mineral and acts as a cofactor in numerous metabolic processes. There is evidence that specific nutrients, such as magnesium, may help to promote muscle relaxation and restfulness—possibly playing a role in reducing daily stress levels.21 Conclusion However you are feeling in any given moment is okay. As humans, we experience a multitude of emotions that change throughout the day. If you’ve noticed you haven’t felt yourself lately, remind yourself that every day is a new day—filled with opportunities for growth and change. Be kind to yourself, use the mantras within this article or create your own that speak to you and consider adding the supportive nutrients listed above to your routine if you are seeking nutritional support for your emotional state. References: 1. Bailenson J. Technology, Mind, and Behavior. 2021;2:1. 2. Beggiato S et al. Front Pharmacol. 2019;10:821. 3. PubChem, U.S. National Library of Medicine, National Center for Biotechnology Information. https://pubchem.ncbi.nlm.nih.gov/compound/4671. Accessed September 20, 2019. 4. Passavanti MB et al. Syst Rev. 2019;8(9). 5. Hesselink JM et al. Int J Inflamm. 2019;2013(9). 6. Hesselink JMK. J Pain Res. 2013;6:625–634. 7. Tagne AM et al. Pharmacol Res. 2021:105545. 8. Hwang TJ et al. Int Psychogeriatr. 2020;32(10):1217-1220. 9. Kim H et al. Prog Lipid Res. 2014;56:1-18. 10.Benton D et al. Nutr Neurosci. 2001;4(3):169-178. 11. Heymsfield S et al. Int J Obes Relat Metab Disord. 2003;27(5):537-549. 12. Cohen MM. J Ayurveda Integr Med. 2014;5(4):251-259. 13. Mirjalili MH et al. Molecules 2009;14:2373–2393. 14. Mindell E. New York, NY, Hachette Book Group, 2011. 15. Panossian AG et al. Med Res Rev. 2021;41(1):630-703. 16. Panossian A. Pharmaceuticals. 2010;3:188-224. 17. NIH Office of Dietary Supplements. Office of Dietary Supplements – Vitamin B12. https://ods.od.nih.gov/factsheets/VitaminB12-HealthProfessional/ . Accessed: August 5, 2021. 18. NIH Office of Dietary Supplements. Office of Dietary Supplements – Folate. https://ods.od.nih.gov/factsheets/Folate-HealthProfessional/ . Accessed: August 5, 2021. 19. Young SN. J Psychiatry Neurosci. 2007;32:394-399. 20. Dakshinamurti K. Adv Nutr Res. 1982;4:143-179. 21. de Baaij JH, et al. Physiol Rev. 2015; 95:1-46. |
Categories
All
Archives
April 2024
|
|
Join Our Community
|
|
Amipro Disclaimer:
Certain persons, considered experts, may disagree with one or more of the foregoing statements, but the same are deemed, nevertheless, to be based on sound and reliable authority. No such statements shall be construed as a claim or representation as to Metagenics products, that they are offered for the diagnosis, cure, mitigation, treatment or prevention of any disease. |













 RSS Feed
RSS Feed