|
Estrogen is a vital hormone that helps support the reproductive organs. Its metabolism is linked to diet, lifestyle, and genetic makeup.
Just like our bodies, our surroundings too carry varying degrees of estrogen. Plastic containers, nail polish, receipts—our households feature more of the hormone than most people know. And while the body can absorb these environmental estrogens (also known as xenoestrogens) in the same way it can produce estrogen naturally, it’s important to maintain a sense of balance. This is because estrogen dominance—that is, the dominance of estrogen over progesterone—can lead to undesired health effects.1,2 This is where an environmental estrogen detox may serve you. There are a number of steps you can take to help reduce the amount of estrogen in your environment:
These are just some of the steps you can take to help lower estrogen levels in your environment and body. Note that an estrogenic detox is best completed by making small, conscious changes. References
Submitted by the Metagenics Marketing Team
0 Comments
The female-centric 411 on this essential nutrient
by Ashley Jordan Ferira, PhD, RDN Overview Vitamin D research and daily news headlines are ubiquitous. PubMed’s search engine contains over 81,400 articles pertaining to vitamin D.1 Information abounds on vitamin D, but the vetting and translation of that information into pragmatic recommendations is harder to find. Evidence-based takeaways and female-centric recommendations are crucial for healthcare practitioners (HCPs), their female patients and consumers alike. Women are busy, multi-tasking pros, so practical, personalized takeaways are always appreciated. In other words, women need the “411” on vitamin D. Merriam-Webster defines “411” as “relevant information” or the “skinny”.2 So for all of you busy women, here’s the skinny on vitamin D. Let’s explore common questions about this popular micronutrient. Q: Is vitamin D more important for younger or older women? A: All of the above. Vitamin D plays a critical role in women’s health across all life stages, from fertility/conception, to in utero, childhood, adolescence, adulthood, older adulthood, and even in palliative care. Vitamin D is converted by the liver and kidneys into its active hormone form: 1,25-dihydroxyvitamin D. This dynamic hormone binds nuclear receptors in many different organs in order to modulate gene expression related to many crucial health areas across the lifecycle, including bone, muscle, immune, cardiometabolic, brain, and pregnancy to name a few.3 Q: I am a grandmother. Are my vitamin D needs different than my daughter and granddaughter? A: Yes, age-specific vitamin D recommendations exist. As an essential fat-soluble vitamin, women need to achieve adequate levels of vitamin D daily. Age-specific Recommended Dietary Allowances (RDA) from The Institute of Medicine (IOM),4 as well as newer clinical guidelines from The Endocrine Society,5 provide helpful clinical direction for daily vitamin D intake and/or supplementation goals. The IOM RDAs4 are considered by many vitamin D researchers to be a conservative, minimum daily vitamin D intake estimate to support the bone health of a healthy population (i.e. prevent the manifestation of frank vitamin D deficiency as bone softening: rickets and osteomalacia): Infants (0-1 year): 400 IU/day Children & Adolescents (1-18 years): 600 IU/day Adults (19-70 years): 600 IU/day Older Adults (>70 years): 800 IU/day The Endocrine Society’s clinical practice guidelines5 recommend higher daily vitamin D levels than the IOM, with a different end-goal: raising the serum biomarker for vitamin D status [serum 25-hydroxvitamin D: 25(OH)D] into the sufficient range (≥ 30 ng/ml) in the individual patient: Infants (0-1 year): At least 1,000 IU/day Children & Adolescents (1-18 years): At least 1,000 IU/day Adults (19+ years): At least 1,500 – 2,000 IU/day Q: I am a health-conscious woman who eats a nutritious, well-rounded diet. I should not need a vitamin D supplement, right? A: Not so fast. Daily micronutrient needs can be met via diet alone for many vitamins and minerals. Vitamin D is one of the exceptions, which is why an alarming number of Americans (93%) are failing to consume the recommended levels from their diet alone.6-7 Very few foods are endogenous sources of animal-derived vitamin D3 (cholecalciferol) or plant-derived vitamin D2 (ergocalciferol). Some natural vitamin D sources include certain fatty fish (e.g. salmon, mackerel, sardines, cod, halibut, and tuna), fish liver oils, eggs (yolk) and certain species of UV-irradiated mushrooms.8 In the early 20th century, the US began fortifying dairy and cereals with vitamin D to help combat rickets, which was widespread. For example, one cup (8 fluid ounces) of fortified milk will contain approximately 100 IU of vitamin D. Even though some food sources do exist, the amounts of these foods or beverages that an adult would need to consume daily in order to achieve healthy 25(OH)D levels (> 30 ng/ml) is quite unrealistic and even comical to consider. For example, you would need to toss back 20 glasses of milk daily or 50 eggs/day to achieve 2,000 IU of vitamin D! In contrast, daily vitamin D supplementation provides an easy and economical solution to consistently achieve 2,000 IU and any other specifically targeted levels. Q: I enjoy the outdoors and get out in the sun daily, so I should be getting all of the vitamin D that I need, correct? A: Vitamin D is a highly unique micronutrient due to its ability to be synthesized by our skin following sufficient ultraviolet (UV) B irradiation from the sun. Many factors can result in variable UV radiation exposure, including season, latitude, time of day, length of day, cloud cover, smog, skin’s melanin content, and sunscreen use. Furthermore, medical consensus advises limiting sun exposure due to its established carcinogenic effects. Interestingly, even when dietary and sun exposure are both considered, conservative estimates approximate that 1/3 of the US population still remains vitamin D insufficient or deficient.9 Q: What factors can increase my risk for being vitamin D deficient? Are there female-specific risk factors? A: Although the cutoff levels for vitamin D sufficiency vs. deficiency are still debated amongst vitamin D researchers and clinicians, insufficiency is considered a 25(OH)D of 21-29 ng/ml, while deficiency is < 20 ng/ml.5 Therefore, hypovitaminosis D (insufficiency and deficiency, collectively) occurs when a patient’s serum 25(OH)D falls below 30 ng/ml. The goal is 30 ng/ml or higher. Ideally, vitamin D intake recommendations4-5 and therapy are personalized by the HCP based on patient-specific information, such as baseline vitamin D status, vitamin D receptor single nucleotide polymorphisms and other pertinent risk factors. Common risk factors for vitamin D deficiency to look out for include: -Overweight/obesity -Older age -Regular sunscreen use -Winter season -Frequent TV viewing -Dairy product exclusion -Darker skin (more melanin) -Not using vitamin D supplements -Malabsorption disorders (e.g. bariatric surgery, IBD, cystic fibrosis) -Liver disease -Renal insufficiency -Certain drug classes: weight loss, fat substitutes, bile sequestrants, anti-convulsants, anti-retrovirals, anti-tuberculosis, anti-fungals, glucocorticoids -Lastly, additional female-specific risk factors to look out for include exclusive breastfeeding while mother is vitamin D insufficient (can result in infant being vitamin D deficient) and certain cultural clothing that covers significant amounts of skin surface area (e.g. hijab, niqab). Key Takeaways
Ashley Jordan Ferira, PhD, RDN is Manager of Medical Affairs and the Metagenics Institute, where she specializes in nutrition and medical communications and education. Dr. Ferira’s previous industry and consulting experiences span nutrition product development, education, communications, and corporate wellness. Ashley completed her bachelor’s degree at the University of Pennsylvania and PhD in Foods & Nutrition at The University of Georgia, where she researched the role of vitamin D in pediatric cardiometabolic disease risk. Dr. Ferira is a Registered Dietitian Nutritionist (RDN) and has served in leadership roles across local and statewide dietetics, academic, industry, and nonprofit sectors. Food-Fortified Vitamin D3 More Effective than D2 at Raising Serum 25(OH)D Levels | Blog | Metagenics18/7/2019 by Ashley Jordan Ferira, PhD, RDN
A food fortification trial demonstrated that 600 IU of daily vitamin D3 had a significantly greater impact than 600 IU of daily vitamin D2 in elevating serum blood levels of 25-hydroxyvitamin D [25(OH)D].1-2 Vitamin D is essential for skeletal health and many emerging extraskeletal physiological processes, but remains one of the most common micronutrient dietary gaps, resulting in widespread hypovitaminosis D globally. Understanding how much vitamin D the body needs daily, in what form, and from what sources is still being discovered. There are two forms of vitamin D: plant-based ergocalciferol (vitamin D2) and animal-based cholecalciferol (vitamin D3). D2 can be found in UV-irradiated mushrooms, certain fortified foods (breakfast cereals, margarine, and milk), dietary supplements, and vitamin D prescription medications. D3 is found in oily fish, egg yolks, fortified milk, and dietary supplements.4 Chemically, D2 and D3 are almost identical except for key side chain differences, with D2 having an additional double bond. D3 has been shown to have a higher affinity to the vitamin D binding protein, hepatic 25-hydroxylase (enzyme that converts vitamin D to the circulating 25(OH)D form) and vitamin D receptor. Whether these chemical and cellular differences translate into differential abilities in raising serum 25(OH)D, the clinical measure of vitamin D status, has been a hotly debated topic since the early 20th century.5 Research literature to date demonstrates a robust case gaining momentum for vitamin D3 and against vitamin D2 for supplementation. In particular, a 2012 systematic review and meta-analysis by of randomized controlled vitamin D supplementation trials in humans explored a head-to-head comparison of vitamin D2 vs. D3 in raising serum 25(OH)D; vitamin D3 was clearly shown to be more efficacious at raising and maintaining serum 25(OH)D levels than vitamin D2.4 Authors concluded that vitamin D3 may be considered the preferred choice for supplementation.4 Since natural sources of vitamin D (dietary input and UVB exposure from the sun) are limited, and a daily vitamin D supplementation regimen is a personal health decision, vitamin D fortification of the food supply is an important, strategic public health measure to help increase dietary vitamin D intake and improve status in the general population.6 Clarity is needed to elucidate whether D2 and D3 are equally effective sources for food fortification, since both forms are currently utilized in the food supply.6 A study by Tripkovic et al. helps to shed light on key differences.1 Results were published in The American Journal of Clinical Nutrition by Dr. Laura Tripkovic and colleagues from a randomized, double-blind, placebo-controlled food fortification trial that included 335 healthy South Asian and white European women aged 20–64 years.1 Participants were randomized to one of five groups: 1) Placebo: Placebo juice with placebo biscuit 2) D2J: Juice supplemented with 15 mcg vitamin D2 with placebo biscuit 3) D2B: Placebo juice with biscuit supplemented with 15 mcg vitamin D2 4) D3J: Juice supplemented with 15 mcg vitamin D3 with placebo biscuit 5) D3B: Placebo juice with biscuit supplemented with 15 mcg vitamin D3 Fifteen mcg of vitamin D is equivalent to 600 IU of vitamin D, which is the US Recommended Daily Allowance (RDA) for ages 1-70 years.7 The daily food-fortified intervention was 12 weeks long during the winter, and serum total 25(OH)D levels were collected at baseline, week 6 and week 12. Data analysis combined ethnic groups. D3-fortified consumption was shown to be twice as effective as D2 in raising 25(OH)D serum levels in the body.1 While the placebo group experienced a 25% reduction in serum 25(OH)D levels over the course of the study, the D2J and D2B groups saw 25(OH)D increases of 33% and 34%, respectively. Most effective, however, were the D3 groups, with 25(OH)D increases in the D3J and D3B groups of 75% and 74%, respectively. The D3J group induced higher incremental increases in 25(OH)D levels: 16.9 nmol/L higher than the D2J group, 16.0 nmol/L higher than the D2B group, and 42.9 nmol/L higher than the placebo group.1 Both juice- and biscuit-supplemented vitamin D3 groups demonstrated similar results, with no statistical differences seen between D3J and D3B groups.1 Compared to white European women, the South Asian women demonstrated a greater increase in 25(OH)D levels in response to both D2 and D3, which was likely caused by their lower baseline vitamin D status.1 This study shows that modest supplementation levels (600 IU daily) of D3 in food and beverage sources twice as effective at raising serum levels of 25(OH)D than vitamin D2.1 This study and previous supplementation studies may impact future policy and practice for vitamin D supplementation source. Additional research addressing dose response, bioactivity of D3 versus D2 and the impact of foods with high levels of vitamin D3 is needed.2 Why is this Clinically Relevant?
References
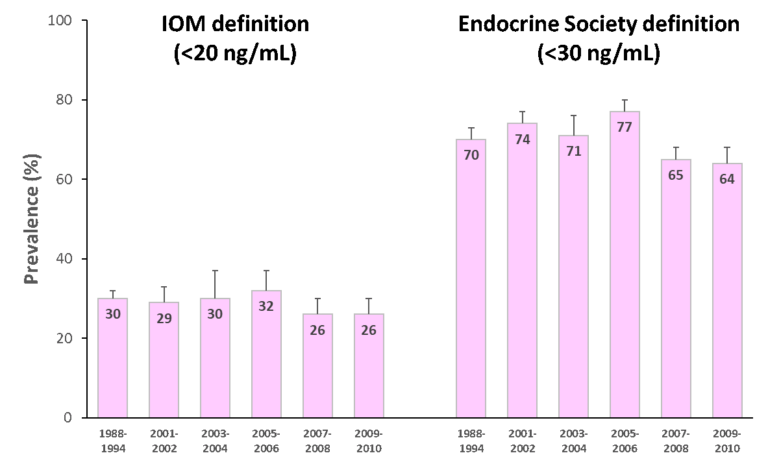
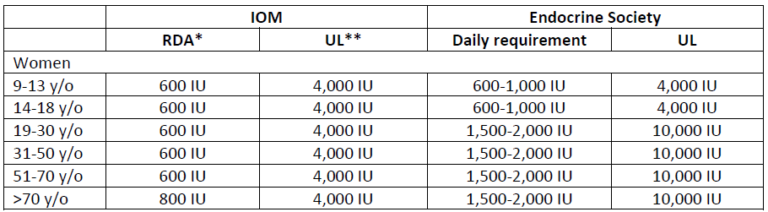
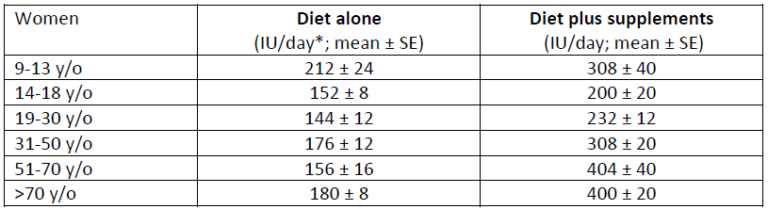
Vitamin D deficiency and the need for practitioner intervention by Sara Gottfried, MD and Lewis Chang, PhD Introduction When I finished my medical training in 1998 at the University of California at San Francisco, I thought I knew everything about vitamin D, particularly vitamin D deficiency and insufficiency. I understood vitamin D was important for the efficient trafficking of calcium in the body, and that helps to keep bones strong, which was important for women’s health over the age of thirty when bone density begins to decline. What I learned in the subsequent twenty years is that vitamin D is both a hormone and a vitamin, and an essential nutrient for more than 400 biochemical jobs in the body. And I learned that what I knew in residency was the tip of the iceberg. Further, I discovered that I inherited a gene for a faulty vitamin D receptor (VDR), which means my body doesn’t absorb and transport vitamin D well, so I tend to have low levels of vitamin D in my blood. This potentially puts me at greater risk for accelerated bone loss, osteopenia, osteoporosis, multiple sclerosis, and certain malignancies such as colorectal cancer. I’m not that unique in carrying the risky gene, but I’m getting ahead of myself. More later on the gene/environment interactions as they relate to vitamin D status, and whether you should be checking your patients for them. As I continue to identify my own knowledge gaps regarding vitamin D, I have become obsessed with teaching other practitioners and my patients about how persistent vitamin D deficiency and insufficiency continue to be despite the public initiatives to raise awareness. In this series on vitamin D, I want to share with you the importance of vitamin D to your health and that of your patients, starting first in this article with how vitamin D status is determined, how prevalent vitamin D insufficiency and deficiency have been in our population, what factors can affect circulating vitamin D levels, recommendations for your female patients since they are at greater risk of problems, and what proven actions to take to help improve your patients’ vitamin D status. How is vitamin D deficiency defined? Serum 25-hydroxyvitamin D [25(OH)D] level is widely used as a bio marker of a person’s vitamin D status, for it is the main circulating form of vitamin D and the best estimator of both endogenous vitamin D synthesis in the skin upon exposure to UV-B light and dietary intake of foods and supplements containing vitamin D.1 Therefore, the diagnosis of vitamin D deficiency is based on the measurement of circulating 25(OH)D levels. However, there is no consensus among key opinion leaders for what is considered an appropriate cutoff for normal, desirable, and optimal serum 25(OH)D concentrations. As a result, different cut-off points have been used to define hypovitaminosis D (deficiency of vitamin D), ranging from 10 ng/mL (25 nmol/L) to 30 ng/mL (75 nmol/L).2-6 There is no consensus among professional societies, either. In 2010, Institute of Medicine (IOM)—renamed in 2016 as the National Academy of Medicine (NAM)—suggested that circulating 25(OH)D levels need to be at least 20 ng/mL (50 nmol/L) to prevent osteomalacia and maintain bone health in at least 97.5% of the population. Therefore, levels at or higher than 20 ng/mL (>50 nmol/L) are defined as vitamin D sufficient. Levels lower than 20 ng/mL (<50 nmol/L) but above 12 ng/mL (>30 nmol/L) are considered vitamin D insufficient and are inadequate for bone and overall health. Levels below 12 ng/mL (<30 nmol/L) are vitamin D deficient which can cause rickets in infants and children and osteomalacia in adults (Figure 1).7 Conversely, believing the IOM recommendations to be too conservative, the Endocrine Society released its clinical practice guidelines in 2011 in which vitamin D sufficiency, insufficiency and deficiency was defined as circulating 25(OH) levels above 30 ng/mL (>75 nmol/L), between 20-30 ng/mL (50-75 nmol/L), and below 20 ng/mL (<50 nmol/L), respectively (Figure 1).8 These recommended cut-off points are similar to the guidelines released by the Central European Scientific Committee on Vitamin D.9 The Endocrine Society also argued that, since any methodology for 25(OH)D measurement is subject to assay variability, targeting a higher 25(OH)D cut-off value may ensure that an individual meets the requirement for sufficient vitamin D levels with only minimal risk of vitamin D toxicity.8 These conflicting professional recommendations result in constant debate among researchers and confusion among clinicians about where the optimal 25(OH)D levels fall. Nevertheless, it is at least agreed upon that serum vitamin D levels less than 30 ng/mL are definitely insufficient and probably deficient for most people. I advise 50-75 ng/mL. Figure 1. 25(OH)D recommendations by the Institute of Medicine (IOM) and the Endocrine Society. How prevalent is vitamin D insufficiency and deficiency? The prevalence varies depending on how vitamin D insufficiency and deficiency is defined. Another important factor that needs to be considered is how 25(OH)D is measured and how differences in the assay methodology (such as assay sensitivity and analytical variability) confound prevalence estimation. Figure 2. Prevalence of vitamin D insufficiency and deficiency (%) from 1988 to 2010 in the U.S. using standardized serum concentrations of 25(OH)D.10Since 1988, the National Health and Nutrition Examination Surveys (NHANES) have periodically tracked nutrition status of adults and children in the U.S. via examining a nationally representative sample population. These estimates are highly valuable in assessing the trends of vitamin D status over time. However, different assays have been used to determine 25(OH)D levels between 1988 and 2010, from DiaSorin radioimmunoassay to reformulated radioimmunoassay to the more accurate chromatography-based assays. To minimize laboratory method bias and imprecision in these different assays, the Vitamin D Standardization Program—initiated by the US Office of Dietary Supplements in the National Institutes of Health in collaboration with the National Institute of Standards and Technology, Centers for Disease Control and Prevention, and Ghent University—was organized in 2010 to promote standardize 25(OH)D measurement using liquid chromatography-tandem mass spectrometry (LC-MS/MS).11 Also, equations were developed to help standardize data from older radioimmunoassays to LC-MS/MS-equivalent data. The first paper that reported the temporal trends of vitamin D status in the U.S. using standardized 25(OH)D measurement was published in American Journal of Clinical Nutrition (AJCN) in 2016. They found that the prevalence of IOM-defined vitamin D insufficiency and deficiency (together as one category) was 30-32 percent from 1988 to 2006 and 26 percent from 2007 to 2010 (Figure 2; left). The prevalence was even higher when the Endocrine Society recommended cut-off point was used: 70-77 percent from 1988 to 2006, dropping slightly to 64-65 percent from 2007 to 2010 (Figure 2; right). Also, non-Hispanic blacks and Mexican Americans had much higher prevalence of vitamin D insufficiency and deficiency than non-Hispanic whites.10 Prior to the development of standardizing 25(OH) measurements, an earlier study also had tracked the prevalence trend in the same NHANES cohort and found an increase in vitamin D deficiency over time.1 Now, from the standardized data, we know the prevalence of vitamin D insufficiency and deficiency has been relatively stable. This discrepancy illustrates the importance of method standardization and having data expressed in LC-MS/MS equivalents for the most accurate interpretation of the prevalence trends. This AJCN report also reported a small increase in vitamin D supplement use (≥ 600 IU/day) among women over time: 2.1% during 1998-1994, increasing to 19% during 2009-2010. Vitamin D supplement users tend to be older (≥40 y/o) and non-Hispanic whites, Still, the majority remained to be non-users; at 70% during 1988-1994 and at 57% during 2009-2010.10 What are the recommended dietary intakes of vitamin D for women? The recommended dietary intakes of vitamin D for women from the IOM and the Endocrine Society are compared in Table 1. IOM recommends 600 IU/day for those who are 9-70 y/o and 800 IU/day for those more than 70 y/o to maintain bone health in at least 97.5% of the population in those age groups. Pregnant and lactating women have the same requirement of 600 IU/day. The intake levels recommended by the Endocrine Society are higher than IOM’s. For those who are 9-18 y/o, at least 600 IU/day is needed. However, in order to consistently achieve 25(OH)D of 30 ng/mL (75 nmol/L) preferred by the Endocrine Society, 1000 IU/day may be required. For those who are 19 y/o and older, including pregnant and lactating women, at least 1500-2000 IU/day may be required to maintain the blood levels of 25(OH)D to above 30 ng/mL (75 nmol/L). Table 1. The recommended dietary intakes of vitamin D for women from IOM and the Endocrine Society. *RDA (Recommended Dietary Allowance): the average daily dietary intake level that is sufficient to meet the nutrient requirement of 97.5% of healthy individuals in a group. **UL (Tolerable Upper Intake Level): the highest level of daily nutrient intake that is likely to pose no risk of adverse health effects to almost all individuals in the general population. Are dietary intakes of vitamin D sufficient in women in the U.S.? Overall no. Even among supplement users, the majority of women have insufficient intakes of vitamin D. Read more in this additional article debunking the myths of vitamin D. The first study that presented national estimates of vitamin D intakes was conducted by the Office of Dietary Supplements and the National Institutes of Health based on NHANES 2005-2006 data. The average vitamin D intake from diet alone was low in every age group (Table 2); the lowest being those 19-30 y/o (144 IU/day) and the highest 9-13 y/o (212 IU/day). When including vitamin D-containing supplement, vitamin D intakes somewhat increased, particularly among those 51 y/o and older (to approximately 400 IU/day).12 Even so, the intake still fell short of the RDA (600-800 IU/day) and way below the levels recommended by the Endocrine Society (1,500-2,000 IU/day). Table 2. Mean vitamin D intake from the diet and from diet plus dietary supplements in women based on NHANES 2005-2006 data.12 *40 IU/day = 1 µg/day The low vitamin D intake seen during 2005-2006 was not a single event. A subsequent study that examined data from NHANES 2001-2002, 2003-2004, 2005-2006, and 2007-2008 confirmed the previous pattern: vitamin D intake from diet only remained extremely low across all populations and age groups, falling short of the RDA even when dietary supplement use was included.13
Further, this study identified certain sub populations that were at greater risks of insufficient vitamin D intakes. When comparing mean intakes by race, African Americans and Mexican Americans had even lower intakes than non-Hispanic whites. When stratifying by annual house income levels, low income was linked to lower vitamin D intakes. Further, those who were affected by obesity had the lowest intake levels compared with those who had normal body weight.13 What about dietary intakes of vitamin D in other countries? Insufficient intakes of vitamin D is not just a phenomenon in the U.S. In fact, it is a global issue. For example, a study involving populations in representative Western countries (Germany, U.K., and the Netherlands) concluded that vitamin D intakes are below recommendations in a significant part of the population in these countries.14 A more recent overview of vitamin D status globally found that vitamin D deficiency occurred all over the world such as in the Middle East, China, Mongolia, and India.15 How much vitamin D intake is needed to improve vitamin D status in women? The Women’s Health Initiative Observational Study (WHI-OS), one of the largest cohorts of postmenopausal women in the U.S., has detailed data on vitamin D intake and serum 25(OH)D level, which has allowed the investigators to address this question. Analysis of the data identified a more or less linear relationship between vitamin D intake and serum vitamin D status: every 100 IU increase in total vitamin D intake (diet and supplement combined) was associated with approximately 0.83 ng/mL (2.08 nmol/L) higher serum 25(OH)D levels. Also, total vitamin D intake was the main factor that determined vitamin D status.16 This dose-response observation is consistent with findings from VIDOS (Vitamin D Supplementation in Older Subjects) study, a 1-year randomized trial that investigated vitamin D intakes and serum 25(OH)D levels in healthy postmenopausal women.17 VIDOS study also found that vitamin D intake at 1,600 IU/day could achieve serum 25(OH)D of 30 ng/mL (75 nmol/L) in 97.5% of the participating women. This dosage level is in line with the recommendations from the Endocrine Society. Is increasing sun exposure alone able to optimize vitamin D status? Some insights can be gained from a study which investigated serum 25(OH)D levels in a group of healthy men who had just completed a summer season of extended outdoor activity such as landscaping, construction work, farming, or recreation. Compared with the general population, these were people who received some of the highest sun exposure—and with sufficient body surface area exposed and without sunscreen—during the season when UV-B radiation was the strongest. The study found that median serum 25(OH)D concentration in these subjects at late summer reached 48.8 ng/mL (122 nmol/L). However, even with ample summer sun exposure, the median serum 25(OH)D dropped to 29.6 ng/mL (74 nmol/L) by late winter, barely meeting vitamin D sufficiency (30 ng/mL) defined by the Endocrine Society.18 Latitude also determines whether one may obtain sufficient levels of vitamin D via sun exposure. One study compared sun exposure in two different cities, Miami (latitude 26 degrees N) and Boston (latitude 42 degrees N), and found that it’s possible to synthesize vitamin D via skin in all months in Miami but it’s very difficult to do so during winter in Boston.19 Therefore, for a very specific group of people such as landscapers, construction workers and farmers who don’t live in high latitude and don’t use sunscreen, their vitamin D level during summer would be sufficient even without dietary intakes of vitamin D. However, their healthy vitamin D status is not sustainable by end of winter if they don’t have additional sources of vitamin D. For the majority of the population—those who spend most of their daylight hours indoors (e.g., office workers), use sunscreen, have their body surface area covered up (e.g., hat, long-sleeve shirt and pants), or live a sedentary lifestyle, etc.—it is highly unlikely that sun exposure alone can increase serum 25(OH)D to sufficient levels even during summer, let alone the rest of the year. What are other important factors affecting vitamin D status? Key non-modifiable factors include age, sex, and race/ethnicity. Although the IOM, the Endocrine Society, and many other professional organizations around the world recommend intake levels based on age and sex, it does not mean that age and sex are the most impactful factors of vitamin D status. Race/ethnicity is another factor that one cannot change, either. However, many factors (besides vitamin D intake) that affect vitamin D status are behavior- and lifestyle-related and therefore modifiable. A 2018 analysis based on NHANES 2001-2010 data has identified several factors and calculated adjusted prevalence ratios of vitamin D deficiency (<20 ng/mL or 50 nmol/L) and insufficiency (20-30 ng/mL or 50-75 nmol/L) in the general population:20
The link between heavier body weight and poorer vitamin D status is a more recent discovery but from the public health viewpoint its impact can be significant, especially when the obesity epidemic has shown no sign of slowing. Due to higher fat mass, a larger individual may simply need more ingestion or internal synthesis of vitamin D to reach the same concentration as a smaller individual.21 This hypothesis is strengthened by findings from a weight loss intervention trial involving postmenopausal women which demonstrated an increase in serum 25(OH)D levels after weight loss.22 The link between physical activity and vitamin D status may be related to other factors. Decreased physical activity level, especially outdoor types, is associated with reduced sunlight exposure which limits vitamin D synthesis from the skin.8Lower physical activity may also contribute to weight gain leading to lower serum 25(OH)D levels due to the effect of dilution. Therefore, increasing outdoor physical activity can improve vitamin D status via increased sunlight exposure and improved body weight. Do genetic factors play an important role in determining vitamin D status? Several genes encode proteins and enzymes that are involved in vitamin D metabolism. Understandably, genetic variations such as single nucleotide polymorphisms (SNPs) of these genes may influence vitamin D status. The first genome-wide association study (GWAS) to identify common genetic variants that influence serum 25(OH)D levels was conducted in 2010 by the SUNLIGHT Consortium involving approximately 30,000 Caucasians from Europe, Canada and the U.S.23 This study identified four SNPs that were associated with lower 25(OH)D levels:
In 2018, the consortium was expanded to nearly 80,000 individuals (all Caucasians) from 31 epidemiological cohorts.28The study confirmed the previous four genetic loci and identified two novel loci for serum 25(OH)D levels:
However, this is an area of active research, and different genetic variations associated with vitamin D status are being discovered in other studies involving other study populations.32-36 Further, little is yet known on how genetic variants interact with other non-genetic factors. For example, one cohort study involving 1,200 postmenopausal women of European descent found that the association between 25(OH)D levels and certain SNPs in GC and CYP2R1 genes were stronger in summer months but not winter months, or in individuals with dietary intakes of vitamin D above 400 IU/day but not below.37 Also, a randomized controlled trial involving nearly 1,800 adults demonstrated that the increase in 25(OH)D after consumption of 1,000 IU/day vitamin D for 1 year was modified by certain SNPs in CYP2R1, CYP24A1, and VDRgene.38 These gene-environmental interactions and associations, once repeated in larger epidemiological studies and confirmed in large clinical trials, will have important public health implications. It will help determine whether individuals with certain genetic risk factors may indeed require higher amounts of vitamin D to achieve sufficient levels of 25(OH). Institute of Medicine miscalculation leads to the wrong recommendation of 600 IU/day As we wrap up, I would like to share with you an interesting article, published in 2014, which was titled “A Statistical Error in the Estimation of the Recommended Dietary Allowance for Vitamin D.” (I have included the hyperlink here in case you are interested in reading the whole open-access article.) In brief, researchers from School of Public Health, University of Alberta (Canada) described how IOM’s RDA for vitamin D was underestimated due to a calculation error. IOM estimated that 600 IU/day of vitamin D would achieve serum 25(OH)D above 20 ng/mL (50 nmol/L) for 97.5% of the population, but the authors re-examined the data and estimated that 600 IU/day of vitamin D would only achieve 10.7 ng/mL (26.8 nmol/L) in 97.5% of the population.39 This means that the dose needed to achieve 20 ng/mL (50 nmol/L) in the majority of the population would have been significantly much higher than 600 IU/day. Following the publication, another group of researchers verified in another publication that IOM indeed had made a calculation error.40 Miscalculated RDA for vitamin D can have serious public health and clinical implications as the true vitamin D insufficiency and deficiency would have been severely underestimated based on the IOM definition. Many researchers are now calling for the IOM and public health authorities to redefine RDA for vitamin D (closer to what the Endocrine Society has proposed) to allow for appropriate public health and clinical decision-making. In the meantime, I recommend dosing vitamin D to achieve serum sufficiency by starting with the Endocrine Society guidelines, not IOM. In the next series of articles, I will discuss how vitamin D status is related to women’s health, including current scientific data on the genetic influence of different disease outcomes, as well as common clinical issues we face as clinicians on the front lines. References
Sara Gottfried, MD is a board-certified gynecologist and physician scientist. She graduated from Harvard Medical School and the Massachusetts Institute of Technology and completed residency at the University of California at San Francisco. Over the past two decades, Dr. Gottfried has seen more than 25,000 patients and specializes in identifying the underlying cause of her patients’ conditions to achieve true and lasting health transformations, not just symptom management. Dr. Gottfried is the President of Metagenics Institute, which is dedicated to transforming healthcare by educating, inspiring, and mobilizing practitioners and patients to learn about and adopt personalized lifestyle medicine. Dr. Gottfried is a global keynote speaker who practices evidence-based integrative, precision, and Functional Medicine. She recently published a new book, Brain Body Diet and has also authored three New York Times bestselling books: The Hormone Cure, The Hormone Reset Diet, and Younger. Lewis Chang, PhD is Scientific Editorial Manager of R&D at Metagenics. Dr. Chang received his PhD in Nutritional Sciences at University of Washington, along with his MS in Nutrition and Public Health from Teachers College, Columbia University and BS in Pharmacy from National Taiwan University. Prior to joining Metagenics, he conducted dissertation research and completed a research assistantship and postdoctoral fellowship at the Fred Hutchinson Cancer Research Center in Seattle, WA. Dr. Chang has authored or co-authored and managed the publication of over 30 peer-reviewed journal articles and numerous scientific abstracts and posters. He has quite a green thumb, enjoys opera, theater and jazz, and loves cooking, collecting art, and learning to play gypsy jazz guitar. We sat down with Joel Evans, MD to talk about Functional Medicine options for common women’s health issues. This is the first in a series.
Before we get started, can you tell me about your background? My background really is one of a conventionally trained OB-GYN. As I became interested in helping my patients in every way I could, it became clear to me that all I was trained in simply wasn’t enough. The best example of this was a 26-year-old woman who I had the misfortune of diagnosing with breast cancer. She asked: Does it matter what I eat? The answer I gave was, “I don’t know; ask your oncologist.” Her oncologist was very well known, trained at Harvard, and the answer came back: “It doesn’t matter what you eat.” She asked me if I believed that, and I said, “Let’s find out for sure. Let’s have you meet with a nutritionist.” She met with a nutritionist, who said you must avoid all sugar; sugar goes directly to cancer cells. I went to the medical library to do some research on sugar and cancer. I was shocked to discover that there were over 10,000 papers on cancer and sugar! It was at that point that I realized that there’s more to taking care of people than is taught in medical school. That’s when I learned about Functional Medicine and met Jeff Bland and started speaking for Metagenics way, way back in 2002 or 2003. What kinds of patients do you typically see? I see patients that I’ve seen forever as an OB-GYN who come to me for GYN care. Then I see patients that come to me because they want a Functional Medicine doctor. I also see the most complicated issues that are referred to me by my colleagues and healthcare practitioners that have been through IFM that know that I have a level of expertise that justifies the referral. The patients you said you’ve seen forever, do you help guide them into the Functional Medicine realm? Or are they just there for their usual visits? I always try to guide them into the Functional Medicine realm. When I do their routine blood work, it is far more expansive than that ordered by a traditional doctor. I look for the subtleties in thyroid function. I look for subtleties in adrenal function. I ask probing questions like, How’s your mood? How’s your energy? How are you sleeping? By asking those broader questions about issues that most OB-GYNs don’t inquire, I can then target the blood work to address them. When I go over the results, my patients are getting Functional Medicine care without knowing the term. Are they open to that? Most are open to that. There are very few, maybe 2 or 3% that aren’t. The vast majority are, which is why they refer their friends and relatives to see me. I’m able to help them in all areas of their health. How would treatment for fibroids and endometriosis differ from traditional medicine to lifestyle medicine? For example, what tests do you use, and can you manage these conditions with lifestyle adjustments?This is a big question to answer! The treatments in conventional medicine for fibroids and endometriosis are treatments that most patients don’t want. If fibroids are diagnosed during a routine exam, and there are no symptoms, the conventional treatment is to do nothing. Most patients aren’t happy with that. From a Functional Medicine perspective, there are a lot of things that we can do to prevent them from growing and becoming symptomatic. Rarely do you find endometriosis without symptoms other than if you’re doing a pelvic exam and you feel a cyst, get an ultrasound on the cyst, and find out that it has qualities that are consistent with endometriosis. In that case, the only conventional interventions are birth control pills or surgery to get that cyst removed and see if there are other endometriosis lesions in the pelvis, where they can be destroyed with laser or electrocautery. If patients have symptoms of fibroids, it can be a feeling of fullness, it can be abnormal bleeding, or it can be pelvic pain. There really are no long-lasting conventional treatments for fibroids other than some type of surgical procedure done by radiologists where they block the blood flow to the uterus or block the blood flow to the fibroids and the fibroids shrink. What I do, the Functional Medicine approach, is totally different. I looked at the underlying driver of fibroid and endometriosis growth, and that has to do with estrogen. The Functional Medicine approach ensures there is not too much estrogen coming into, or being produced, by the body and that estrogen is being eliminated properly. What are the sources of estrogen entering the body?It can be from foreign substances, so we can be taking chemicals in, compounds that become or behave like estrogen—those are called xeno-estrogens—that’s why eating organic is so important. We can also be drinking estrogen from our water. They have found estrogens from medications in the water. The other way we can have too much estrogen is that we can be producing too much. For example, we can make too much estrogen through an ovarian cyst. Visceral adipose tissue produces estrogen. In addition, we know the enzyme that makes estrogen, aromatase, is actually inside of endometriosis tissue and inside uterine fibroids. Anything that stimulates aromatase will produce more estrogen. Two direct stimulators of aromatase are insulin and inflammatory prostaglandins. Therefore, the Functional Medicine approach includes normalizing insulin levels and reducing inflammation. Another thing to do is look at how the gut functions. If there are unhealthy bacteria in the microbiome, they can produce an enzyme called beta-glucuronidase that leads to reabsorption of estrogen from the intestine. We have to make sure that we move our bowels regularly, because if there’s constipation, that leads to increased estrogen absorption. We also have to make sure our liver is functioning well, that our cytochrome P450 detoxification enzymes are functioning well because estrogen is eliminated through those pathways. You have a special interest in hereditary breast and ovarian cancer. What kind of risk assessment and prevention tools do you use with your patients?There are standard questions that are put out by National Cancer Institute and the National Institute of Health that we use to find out if patients are at increased risk for BRCA. Then there are other questions that we ask. Dense breasts put a woman at increased risk for breast cancer. I also look at risk factors for other hereditary cancer syndromes, such as Lynch syndrome, where women are at increased risk of colon and gynecologic cancer. I also look for other, nongenetic reasons patients might be at increased risk for breast cancer, like being overweight, having increased adipose tissue, elevated insulin levels, or even being under stress. I then also do a test called BREVAGen™, which can give patients a five-year risk analysis of breast cancer. That’s what’s called a genomewide association study. By looking at the gene frequency of approximately 77 genes, they can come up with a very accurate five-year risk of breast cancer. That means they are candidates for risk reduction medications in addition to a thorough Functional Medicine breast cancer risk reduction protocol. Methods to Help Manage Healthy Estrogen Balance
Hormonal balance is complicated. As women, we have a constant ebb and flow of hormones in our bodies that can greatly affect how we feel from day to day. Not only that, these hormones (estrogen, progesterone, and testosterone) work synergistically in the body to keep things running smoothly and hinge on a delicate balance that can easily be disrupted by lifestyle habits and environmental exposures. When that happens, we tend to feel, well, not quite right. Estrogen dominance is the most common type of hormone imbalance—characterized by frequent headaches, mood swings and anxiety, bloating and weight gain, irregular periods, trouble sleeping, unexplained fatigue, worsened PMS symptoms, and more.1 Some of these symptoms may sound familiar to you. Maybe you’ve already been checked for estrogen dominance or hormonal imbalance. The good news is, there are a few things you can do to help manage estrogen dominance—starting today.
Living with estrogen dominance does not have to be a lifelong challenge. With these complementary methods, you can limit your exposure to xenoestrogens and help regulate your body’s natural hormonal balance. If you have not had your hormone levels checked and show signs of estrogen dominance, please visit your healthcare practitioner. *These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.References:
By Nilima Desai, RD
If you have ever experienced hot flashes, night sweats, etc., due to menopause, you are not alone. About 80% of menopausal women suffer from hot flashes, night sweats, sleep disturbances, depression, anxiety, and vaginal dryness,¹ which can significantly affect their quality of life. Menopause is characterized by a decrease in estrogen levels, which triggers these uncomfortable symptoms. Most women report hot flashes to be the most bothersome symptom and the reason for starting hormone therapy.² Symptom Relief OptionsIn addition to lifestyle recommendations, such as following a plant-based diet, increasing physical activity, and minimizing smoking and alcohol intake, the addition of hormone replacement therapy (HRT) has been most effective in reducing vasomotor symptoms (hot flashes, night sweats) commonly associated with decreased estrogen levels.²,³* However, current recommendations from the American Congress of Obstetricians and Gynecologists suggest limiting HRT to the lowest effective dose for the shortest amount of time possible.⁴ As a result, 40-50% of women choose to use practical alternative options, such as plant-derived solutions to address menopause-related symptoms.³ Various plant-derived solutions including phytoestrogens such as isoflavones, lignans, and other Chinese and herbal remedies such as ginseng, black cohosh, etc., have been studied for the relief of menopausal symptoms.* Plant-Derived Solutions: Phytoestrogens are a group of nonsteroidal plant-derived compounds with estrogen-like properties. The chemical structure contains a phenolic ring that enables them to bind to estrogen receptors in the body.⁵ They bind to both types of estrogen receptors, Erα and Erβ.⁵ However, research suggests that majority of the phytoestrogens have a higher affinity to bind to Erβ as compared to steroidal estrogens.⁵ Therefore, they may exert their actions through different pathways and may potentially induce different beneficial responses.* There are four classes of phytoestrogens: isoflavones, lignans, coumestans, and stilbenes.⁶
Results from 21 randomized controlled trials (RCTs) examining the association between different phytoestrogens and menopausal symptoms (frequency and duration of hot flashes, vaginal dryness, etc.) included in a meta-analysis concluded that there was an association of overall phytoestrogen use with a decrease in the number of daily hot flashes and in vaginal dryness scores.³ However, the use of phytoestrogens was not associated with significant changes in 24-hour night sweat episodes.³* ERr 731® is a standardized extract of Siberian rhubarb root, a plant-derived, nonhormonal therapy designed to alleviate menopausal symptoms, including hot flashes.* In a confirmatory RCT involving 119 perimenopausal women, compared with perimenopausal women receiving placebo, those receiving ERr 731® experienced a median 83% decrease in daily hot flashes over the course of 12 weeks.⁷ Compared to placebo, perimenopausal women who received ERr 731® (the extract found in Estrovera) experienced a decrease in symptoms (as indicated by an average [mean] reduction) of up to 83% in individual Menopause Rating Scale scores.⁸ Clinical benefits of ERr 731® appear to be related to selective binding of Erβ and lack of affinity for Erα.9,10* Black cohosh is an herb that has a long history of use for the relief of menopausal symptoms, including hot flashes and night sweats. Results from four RCTs examining the association between black cohosh with menopausal symptoms included in a meta-analysis concluded that black cohosh was not associated with changes in the number of hot flashes and night sweats within a 24-hour period.³ Therefore, although black cohosh is a popular herbal remedy to address menopausal symptoms, research has shown no significant association between black cohosh supplementation and relief in menopausal symptoms.³* Other herbs: There aren’t many studies conducted on the associations of Chinese and non-Chinese medicinal herbs with menopausal symptoms. The few RCTs conducted on the various herbs were not consistent and in general didn’t show any association with symptom relief.³* Although many RCTs have been conducted on phytoestrogens and herbal remedies in relation to menopausal symptom relief, further studies are needed to determine potential long-term adverse health effects.* Next StepsUse of HRT needs to be evaluated carefully, and the clinician should assess the risks and benefits associated with prescribing HRT for each individual woman based on her symptoms and personal and family medical history. For women who choose to avoid or have contraindications to HRT, plant-derived therapies in conjunction with a patient-centered approach may potentially provide an alternative in relieving certain symptoms associated with menopause. To determine the best options, patients should always consult with their healthcare provider. *These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.References:
Many of us (men and women) have had moments where we sneezed and lost control of our bladder, or we had a coughing attack and sprang a little “leak.” But for women in menopause, urinary leakage can occur more frequently and cause embarrassment and concern. Urinary incontinence is actually common during this time in a woman’s lifespan and can be the result of one or more causes. Fortunately, there are many ways to address this problem.
What is urinary incontinence?Simply put, urinary incontinence is the involuntary leakage of urine.1 Up to one-third of men and women in the US have urinary incontinence,1 but it is older women—those going through perimenopause and menopause—who most often develop stress urinary incontinence (SUI).1 Urge incontinence (UI) is also common in postmenopausal women; also known as overactive bladder (OAB), the condition results in a fast, urgent need to urinate, accompanied by urine leakage.2 Bend. Lift. Leak.As time marches on, children are born, and estrogen levels drop, the pelvic floor muscles that support the bladder and other organs can naturally weaken and become stretched. Less support means less ability to hold urine, and when the urethra (the duct where urine comes out of the body) is challenged by a good sneeze or lifting a box, it can leak out a few drops, or up to a tablespoon or more.1 This is SUI, and a decline in estrogen levels plays an important role in its development during menopause, at which time the tissues of the urogenital tract can weaken and become thin.3,4 Additionally, giving birth multiple times, or traumatic vaginal birthing, can contribute to SUI in menopause.4 Gotta go—now!Urge incontinence (UI) is the sudden urge to urinate, often accompanied by urine leakage. In addition to the weakening of pelvic tissues during and after menopause, UI may be caused by a lack of coordination between the brain and bladder, with the signal to urinate being sent by the brain at the wrong time.5Other symptoms of UI include urinating more than eight times per day and more than once at night (nocturia).5 What can be done about urinary incontinence?There are several ways to help make it easier to deal with urinary incontinence. You may start by refraining from alcohol and caffeine; stopping drinking liquids close to bedtime; wearing a disposable pad designed to absorb urine; and maintaining a healthy weight. There are also more comprehensive methods available to address urinary incontinence. Pelvic floor strengthening exercise--The beloved Kegel exercise is a well-known way to help strengthen pelvic floor muscles and possibly eliminate bladder leakage.6 By contracting and relaxing these muscles repeatedly, they can be strengthened and provide better bladder support. Where are your pelvic floor muscles? If you can stop urinating midstream, you’ve found them.6 Ask your healthcare practitioner for information on how to perform the Kegel exercise. Pessary--This small device is inserted into the vagina to help support the pelvic organs and is also used to treat urinary incontinence. Fitted by your healthcare practitioner, a pessary is removable, minimally invasive, and discreet. There are also over-the-counter, disposable bladder supports available that are self-fitting.7,8 Biofeedback--Small sensors are discreetly placed on the body and used to measure what the pelvic floor muscles are doing, as well as provide feedback to help teach you how to control the pelvic floor muscles.9 Medication--Drugs that are designed specifically for urinary incontinence can be used alone or with a method such as biofeedback; they work primarily by relaxing the bladder muscle and increasing the amount of urine the bladder can hold.10 Surgery--While surgery is more invasive than other ways that address urinary incontinence, it can also successfully resolve it. Procedures such as sling surgery and bladder neck suspension can help keep the urethra from moving down and opening accidentally.11 No matter which method you wish to try, always ask your healthcare practitioner about which is right for you. He or she knows your personal health history best and can help you find relief from menopausal urinary incontinence. This information is for educational purposes only. This content is not intended as a substitute for professional medical advice, diagnosis, or treatment. Individuals should always consult with their healthcare professional for advice on medical issues. References:
Interview with Lyra Heller, MA Our bodies change as we age—as hard as we might fight it. For women who want to maintain a sexually active life with a partner, that may mean accommodating some physical changes. We talked to Lyra Heller, MA to learn her advice on what we can do in order to keep that libido alive. How does menopause affect sex? This is a complex question because women’s sexuality is complex. Part of your experience around sexuality and your sexual response is governed by changing hormones. The major impact centers on what menopause means to you, what it symbolizes, your relationship with your partner, and your general health and sense of wellbeing. How do changing hormones affect sex? Menopause is a process. It’s a major life transition marked by declines in sex hormones that signal the end of your reproductive years. Perimenopause is the first phase. It can begin sometime in your 40s and may extend into your mid-50s. The symptoms are related to fluctuating hormone levels that cause changes in menstrual cycle quality and frequency, hot flashes, spontaneous sweats leading to poor sleep quality, anxiety, and moodiness. You may notice some weight gain. Desire to engage in sex can be the last thing on your mind because you are tired, uncomfortable, possibly self-conscious. As estrogen levels continue to decline, vaginal dryness, which loosely translates as you don’t lubricate as well when you’re sexually aroused, may become an issue. This can result in painful intercourse, and it can produce a sense of negativity as you approach sex, because it hurts. Part of the menopausal experience is the vaginal lining tends to thin, and sometimes the walls of your vagina can narrow, so intercourse in general can be hurtful. How do your feelings and thoughts about menopausal changes affect sex? Christiane Northrup, author of Women’s Bodies, Women’s Wisdom: Creating Physical and Emotional Health and Healing says, “Thoughts are an important part of your inner wisdom, and they are very powerful. A thought held long enough and repeated often enough becomes a belief. A belief then becomes your biology. ”1 Menopause is a time of physical transformation that encompasses the aging process. Desiring an active sex life at times requires engaging in difficult and rewarding conversations with yourself and your partner because your body may not perform as before. Sounds simple enough, but it can be challenging to communicate “what turns you on.” Yet this may become a core issue. Other considerations that dampen sexual desire are:
Aging seems to come with increased aches and pains, whether from arthritis or just general physical deterioration. How can pain affect a healthy sex life? Pain hurts a healthy sex life! You don’t want to have sex when you hurt. So what happens is, you decide to control the pain. And there are pain medications that will actually reduce your desire to have sex. In fact, a lot of medications can cause sexual problems. Plus, drug combinations and mixtures of prescription with over-the-counter (OTC) medications are all capable of inducing disinterest in sex. This is where lifestyle becomes really important. If you’re suffering from a chronic health condition, the trajectory of seeking relief should start with a self-focused approach in partnership with your healthcare practitioner. Some of the major issues requiring medications that might affect your sex life can be elevated blood pressure, depression, anxiety, gut problems, and others2—these are all things that can in some instances also be helped by diet and lifestyle and becoming more physically active. With minor health concerns, a healthcare practitioner can provide guidance on what lifestyle modifications may help. Think in terms of being more proactive in how you approach your food choices, how you want to deal with the excess burden of weight, how you want to deal with blood pressure, adrenal function, and elevated blood fats—these are all capable of being modified by a healthy lifestyle. If you have had a heart attack or have coronary artery disease, do you need to be concerned with continuing normal sexual activities? Typically there is no concern as long as there is doctor oversight. If you experience shortness of breath, can’t walk very far, have poorly controlled blood pressure, those kinds of things are going to affect sexual vitality. That said, cardiovascular disease is the leading cause of death for women.3 It is important to appreciate your heart disease risk may go unrecognized even though knowledge about gender differences grows.4 New research suggests that women experiencing hot flashes before age 42 may have an elevated risk of cardiovascular disease when compared to women with late onset vasomotor symptoms (older than 42).5So heart disease is not restricted to women over 65. A baby aspirin a day may not be enough protection.6 Take your heart health seriously. If you are under the age of 65, and especially if you have a family history of heart disease, pay close attention to heart disease risk factors. The risk factors for heart disease are the same as for premature estrogen decline associated with early perimenopause: smoking, physical inactivity, overweight, standard American diet.7 Be proactive. Talk with your doctor. Are there any options out there that can help with libido?Experiencing perimenopause and libido is different from experiencing postmenopause and libido. Perimenopause is a rollercoaster ride of fluctuating hormones. Hot flashes and night sweats reduce your sleep. With the exception of some women whose sex drive may increase during perimenopause, you’re tired or irritable or anxious. If you’re depressed, it may worsen. You may feel old and ugly. As the extreme symptoms subside, if your libido is still hovering around zero, see a doctor to discuss possible interventions. There are several noteworthy methods: vaginal lubricants, moisturizers, and topical hormones. Water-based vaginal lubricants have a short-term effect on dryness. Vaginal moisturizers differ in that they have a longer-term effect and are prescribed on a regular basis—daily or every 2–3 days, depending on the extent of the dryness. Hyaluronic acid vaginal gel may improve symptoms of vaginal dryness, comparable with the effect of topical estrogen therapy. Both are recommended to reduce friction contributing to painful intercourse. Low-dose vaginal topical estriol, a weak estrogen, is an effective way to kindle sexual desire in some women and reduce vaginal dryness. The effect is different from oral hormone replacement therapy (HRT). Topical estriol seems to exert local as opposed to systemic effects. There are other topical hormones available that your doctor can prescribe, as well. If want to be sexually active throughout life, you can. It is a choice—a healthy, rewarding choice. Libido can be nourished. Discovering your capacity for creativity, curiosity, and experimentation is critical to being “turned on.” Grappling with the physical changes of menopause stimulates the need to explore your beliefs and feelings about what is means to be sexually intimate as we age. This can involve venturing into uncharted waters on the adventure of a lifetime. This content is not intended as a substitute for professional medical advice, diagnosis, or treatment. Individuals should always consult with their healthcare professional for advice on medical issues. REFERENCES:
By Nilima Desai, RD If you have ever experienced hot flashes, night sweats, etc., due to menopause, you are not alone. About 80% of menopausal women suffer from hot flashes, night sweats, sleep disturbances, depression, anxiety, and vaginal dryness,¹ which can significantly affect their quality of life. Menopause is characterised by a decrease in estrogen levels, which triggers these uncomfortable symptoms. Most women report hot flashes to be the most bothersome symptom and the reason for starting hormone therapy.²
Symptom Relief OptionsIn addition to lifestyle recommendations, such as following a plant-based diet, increasing physical activity, and minimising smoking and alcohol intake, the addition of hormone replacement therapy (HRT) has been most effective in reducing vasomotor symptoms (hot flashes, night sweats) commonly associated with decreased estrogen levels.²,³* However, current recommendations from the American Congress of Obstetricians and Gynecologists suggest limiting HRT to the lowest effective dose for the shortest amount of time possible.⁴ As a result, 40-50% of women choose to use practical alternative options, such as plant-derived solutions to address menopause-related symptoms.³ Various plant-derived solutions including phytoestrogens such as isoflavones, lignans, and other Chinese and herbal remedies such as ginseng, black cohosh, etc., have been studied for the relief of menopausal symptoms.* Plant-Derived SolutionsPhytoestrogens are a group of nonsteroidal plant-derived compounds with estrogen-like properties. The chemical structure contains a phenolic ring that enables them to bind to estrogen receptors in the body.⁵ They bind to both types of estrogen receptors, Erα and Erβ.⁵ However, research suggests that majority of the phytoestrogens have a higher affinity to bind to Erβ as compared to steroidal estrogens.⁵ Therefore, they may exert their actions through different pathways and may potentially induce different beneficial responses.* There are four classes of phytoestrogens: isoflavones, lignans, coumestans, and stilbenes.⁶
Results from 21 randomized controlled trials (RCTs) examining the association between different phytoestrogens and menopausal symptoms (frequency and duration of hot flashes, vaginal dryness, etc.) included in a meta-analysis concluded that there was an association of overall phytoestrogen use with a decrease in the number of daily hot flashes and in vaginal dryness scores.³ However, the use of phytoestrogens was not associated with significant changes in 24-hour night sweat episodes.³* ERr 731® is a standardized extract of Siberian rhubarb root, a plant-derived, nonhormonal therapy designed to alleviate menopausal symptoms, including hot flashes.* In a confirmatory RCT involving 119 perimenopausal women, compared with perimenopausal women receiving placebo, those receiving ERr 731® experienced a median 83% decrease in daily hot flashes over the course of 12 weeks.⁷ Compared to placebo, perimenopausal women who received ERr 731® (the extract found in Estrovera) experienced a decrease in symptoms (as indicated by an average [mean] reduction) of up to 83% in individual Menopause Rating Scale scores.⁸ Clinical benefits of ERr 731® appear to be related to selective binding of Erβ and lack of affinity for Erα.9,10* Black cohosh is an herb that has a long history of use for the relief of menopausal symptoms, including hot flashes and night sweats. Results from four RCTs examining the association between black cohosh with menopausal symptoms included in a meta-analysis concluded that black cohosh was not associated with changes in the number of hot flashes and night sweats within a 24-hour period.³ Therefore, although black cohosh is a popular herbal remedy to address menopausal symptoms, research has shown no significant association between black cohosh supplementation and relief in menopausal symptoms.³* Other herbs: There aren’t many studies conducted on the associations of Chinese and non-Chinese medicinal herbs with menopausal symptoms. The few RCTs conducted on the various herbs were not consistent and in general didn’t show any association with symptom relief.³* Although many RCTs have been conducted on phytoestrogens and herbal remedies in relation to menopausal symptom relief, further studies are needed to determine potential long-term adverse health effects.* Next StepsUse of HRT needs to be evaluated carefully, and the clinician should assess the risks and benefits associated with prescribing HRT for each individual woman based on her symptoms and personal and family medical history. For women who choose to avoid or have contraindications to HRT, plant-derived therapies in conjunction with a patient-centered approach may potentially provide an alternative in relieving certain symptoms associated with menopause. To determine the best options, patients should always consult with their healthcare provider. *These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.References:
|
Categories
All
Archives
April 2024
|
|
Join Our Community
|
|
Amipro Disclaimer:
Certain persons, considered experts, may disagree with one or more of the foregoing statements, but the same are deemed, nevertheless, to be based on sound and reliable authority. No such statements shall be construed as a claim or representation as to Metagenics products, that they are offered for the diagnosis, cure, mitigation, treatment or prevention of any disease. PAIA Manual |













 RSS Feed
RSS Feed